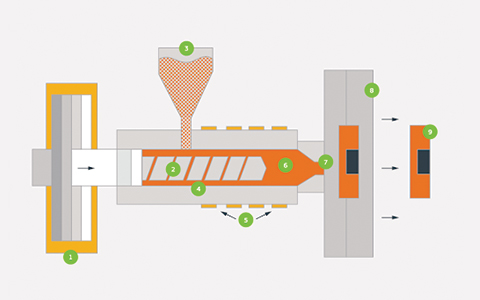
Medical molded parts operate within a regulated framework that extends beyond the molded component itself.
Compliance is determined by legal regulations, quality management systems, material controls, process validation, environmental controls, and documented risk management.
This article explains those requirements using a layered compliance model applicable to medical injection molding suppliers.
A Layered Compliance Framework for Medical Molded Parts
Regulatory compliance for medical molded components can be understood in six layers:
- Legal regulatory framework
- Quality management system
- Design control and risk management
- Material regulatory compliance
- Process validation and manufacturing control
- Environmental and contamination control
Each layer contributes to the regulatory status of the finished medical device.
Layer 1 — Legal Regulatory Framework
Medical devices distributed in the United States are regulated under the Federal Food, Drug, and Cosmetic Act.
The primary regulatory reference for manufacturing is 21 CFR Part 820, now revised as the Quality Management System Regulation QMSR.
Although molded component suppliers are not always classified as finished device manufacturers, they may operate as a medical device contract manufacturing partner supporting OEM regulatory programs.
In these roles, they are subject to supplier audits and documentation review under 21 CFR 820.50.
Under QMSR, FDA requirements align with ISO 13485:2016. However, statutory authority remains with the FD&C Act, and compliance documentation must support inspection readiness.
For OEM customers producing OEM medical components, molded part suppliers must provide records that support:
- Device Master Record inputs
- Device History Record traceability
- Design History File documentation where applicable
This ensures that the component can be integrated into the regulated device lifecycle.
Layer 2 — Quality Management System Requirements
ISO 13485 establishes the global baseline for medical device quality systems.
For regulated medical plastic injection molding, the organization must maintain:
- Documented procedures
- Supplier qualification controls
- Change management processes
- Corrective and preventive action systems
- Risk-based quality planning
ISO 13485 integrates risk management principles and requires validation of special processes.
Injection molding is typically classified as a special process because output cannot be fully verified by inspection alone. Dimensional compliance does not guarantee material integrity or internal stress control.
An ISO-certified molding facility such as SeaSkyMedical integrates molding validation within its quality management system to ensure repeatability and traceability.
Layer 3 — Design Control and Risk Management
Design controls are required under both ISO 13485 and FDA QMSR.
For molded components, this affects tooling strategy, including mold making and early-stage tool design support.
Risk management is commonly aligned with ISO 14971 principles.
Typical molding-related risk considerations include:
- Dimensional drift
- Flash or particulate generation
- Stress cracking
- Seal deformation
- Material degradation after sterilization
During regulated product development, suppliers often support OEM design reviews through:
- Mold flow analysis
- Design for manufacturability evaluation
- Failure mode and effects analysis FMEA
Early prototyping through 3D prototype printing can reduce tooling risk before validation.
Critical-to-quality features must be identified early so that validation focuses on high-risk parameters.
Layer 4 — Material Regulatory Compliance
Material compliance requirements depend on the application and patient contact classification.
Controlled medical material selection is essential to ensure regulatory alignment.
Biocompatibility Standards
ISO 10993 provides the framework for biological evaluation.
Testing requirements vary based on:
- Type of tissue contact
- Duration of exposure
- Route of exposure
Material suppliers may provide baseline biocompatibility data. However, finished product configuration may require additional evaluation.
USP Class VI and Extractables Considerations
USP Class VI testing evaluates biological reactivity under defined extraction conditions.
In regulated programs, molded part manufacturers must maintain:
- Resin lot traceability
- Certificates of analysis
- Controlled storage conditions
Extractables and leachables risk must be evaluated when components contact fluids or drug products.
Certain materials such as LSR injection moulding are selected in applications requiring flexibility and sterilization compatibility.
Additives and color masterbatch must be documented and approved under change control procedures.
Regrind use is typically restricted or prohibited in regulated medical molding unless explicitly validated.
Layer 5 — Process Validation and Manufacturing Control

Regulatory compliance requires validated and controlled molding processes.
Validated plastic injection molding processes must demonstrate repeatability under defined operating windows.
Process Validation
Injection molding validation commonly follows:
- Installation Qualification IQ
- Operational Qualification OQ
- Performance Qualification PQ
Critical process parameters are identified through development studies and design of experiments.
Typical parameters include:
- Melt temperature
- Mold temperature
- Injection pressure
- Holding time
- Cooling time
Validation defines an approved process window that ensures consistent output.
Worst-case conditions are often evaluated to demonstrate process robustness.
Statistical Process Control
Ongoing production typically incorporates statistical monitoring.
Common metrics include:
- Cp and Cpk capability indices
- Control charts for critical dimensions
- Process drift monitoring
Capability targets vary by application but often exceed 1.33 for regulated features.
Secondary Operations and Post-Molding Controls
For devices requiring post-molding operations, controlled secondary operations may include:
These activities must also operate under validated and documented procedures to maintain regulatory integrity.
Layer 6 — Environmental and Contamination Control
Not all medical molded parts require cleanroom production. Requirements depend on application and contamination sensitivity.
Cleanroom Classification
Cleanrooms are classified under ISO 14644.
Common classifications for medical molding include:
- ISO Class 7
- ISO Class 8
Controlled cleanroom injection molding limits airborne particle concentration and supports contamination-sensitive applications.
Contamination Control Practices
Clean manufacturing environments typically implement:
- Segregated medical production areas
- Controlled gowning procedures
- Positive pressure cascades
- Environmental monitoring programs
- Controlled packaging zones
SeaSkyMedical produces medical molded components within an ISO 8 cleanroom environment, supporting applications where particulate control and controlled packaging are required.
Compliance Responsibility Matrix
Regulatory responsibility is distributed across the supply chain.
| Compliance Layer | Primary Responsibility | Supplier Contribution |
|---|---|---|
| Legal compliance | Legal manufacturer | Audit support |
| QMS | Manufacturer and supplier | Certified system |
| Design control | OEM | DFM and tooling input |
| Material validation | OEM | Resin documentation |
| Process validation | Supplier | IQ/OQ/PQ execution |
| Environmental control | Supplier | Cleanroom production |
Clear role definition reduces regulatory risk and supports audit readiness.
Frequently Asked Questions
Do medical molded parts require direct FDA approval
Individual molded components are typically not separately approved by the FDA. However, they must be produced under compliant quality systems that support the finished device’s regulatory status.
Is ISO 13485 mandatory for medical molding suppliers
ISO 13485 is not legally mandatory in all jurisdictions. However, most OEMs require certification because it aligns with FDA QMSR and global regulatory expectations.
What documentation must a molding supplier provide
Common documentation includes:
- Process validation reports
- Material certifications
- Lot traceability records
- Dimensional inspection reports
- Change control records
These documents support the OEM’s regulatory file.
Are cleanrooms required for all medical molded parts
Cleanrooms are required when contamination risk affects device performance or sterility. Structural components inside enclosed devices may not require controlled environments, while fluid-path components often do.
Conclusion
Medical molded parts must comply with a multi-layered regulatory framework that includes legal requirements, quality systems, material controls, validated processes, and environmental controls.
Compliance depends on documented control of materials, validated molding processes, risk-based design evaluation, and traceable production records.
SeaSkyMedical supports regulated manufacturing through certified quality systems, validated medical injection molding processes, and controlled cleanroom production.
For project-specific compliance discussions, contact SeaSkyMedical for more information.