Introduction and Core Definition
Injection molding process validation is a systematic methodology that demonstrates whether a manufacturing process consistently produces parts that meet predetermined quality specifications. Unlike simple first-sample inspection, it establishes repeatability, controllability, and traceability across all production batches. This validation is critical in highly regulated sectors such as medical plastic injection molding, pharmaceuticals, automotive safety components, and defense systems, while also representing best practices for high-quality industrial parts. Proper validation ensures compliance, reduces defects, and safeguards both functionality and safety, while partnering with a capable provider like SeaSkyMedical can streamline this process from design to production.
Regulatory and Quality Standards Framework
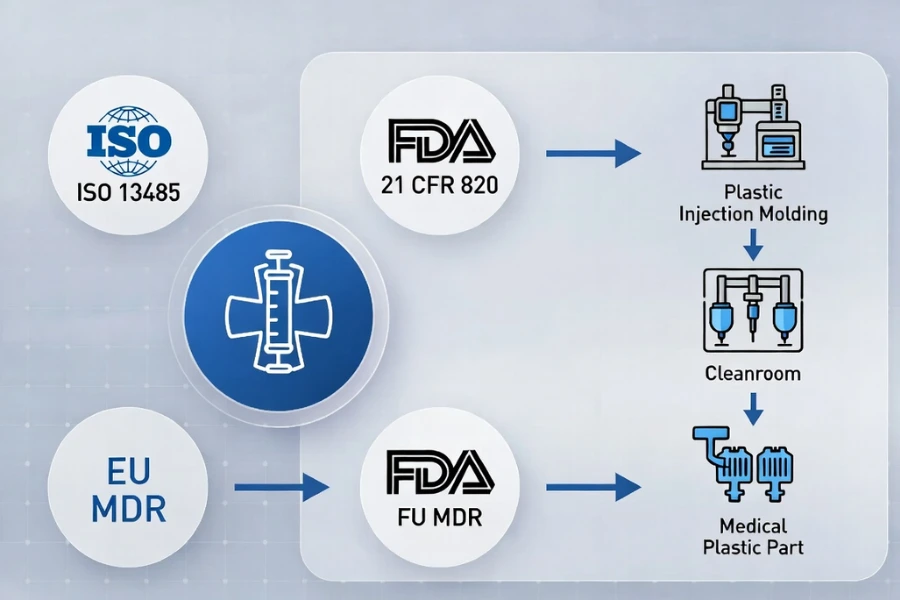
Compliance with multiple regulatory frameworks is the foundation of injection molding process validation. Meeting a single standard is insufficient; manufacturers often integrate several standards to satisfy regulatory and customer expectations.
| Standard/Regulation | Core Requirement | Role in Injection Molding Validation |
|---|---|---|
| ISO 13485 | Medical device quality management system | Requires process validation, risk management, document control, and traceability |
| ISO 10993 | Biological evaluation | Ensures materials are biocompatible for contact with tissue, blood, or skin |
| FDA 21 CFR 820 | Quality System Regulation (GMP) | Validates process and documentation adherence |
| FDA 21 CFR 177 | Plastic materials in contact with drugs/food | Specifies approved resins and labeling requirements |
| EU MDR 2017/745 | Medical Device Regulation | Focus on safety, clinical assessment, post-market surveillance, and UDI traceability |
| ITAR | International Traffic in Arms Regulations | Controls personnel, data, and facility access for defense-related parts |
Compliance is not merely meeting one standard; integration of ISO 13485 with FDA QSR forms the regulatory foundation for medical device contract manufacturing.
Core Validation Framework: IQ / OQ / PQ
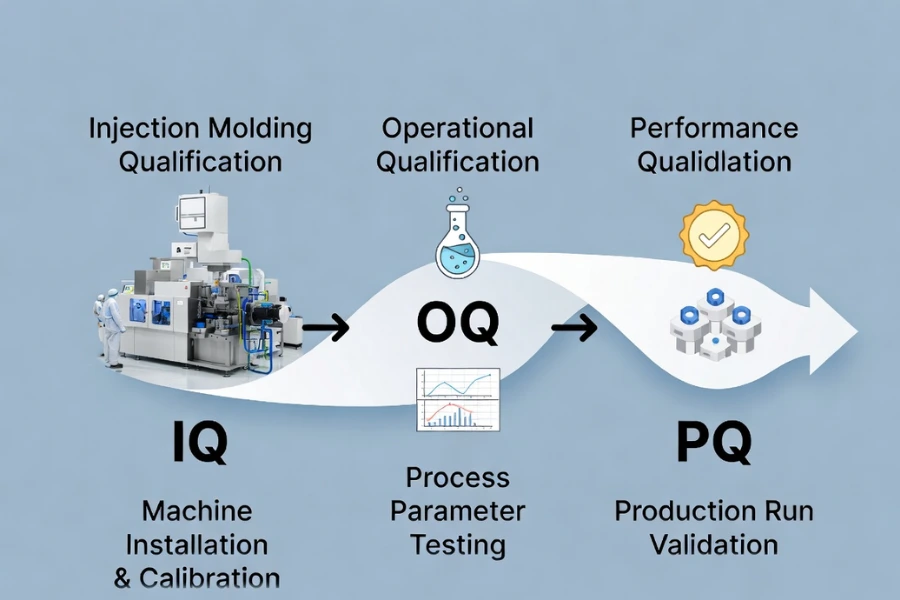
IQ – Installation Qualification
Installation Qualification verifies that injection molding equipment, precision mold making, and auxiliary systems are installed according to design specifications. This includes checking infrastructure, safety mechanisms, electrical and cooling connections, and software configuration. Detailed documentation supports traceability and provides evidence for regulatory audits.
OQ – Operational Qualification
Operational Qualification assesses whether the system operates consistently within defined limits. Critical parameters—melt temperature, mold temperature, injection and hold pressures, cooling times—are tested across upper and lower bounds. CNC plastic machining and DOE methodologies help define the process window and confirm operational stability.
PQ – Performance Qualification
Performance Qualification demonstrates that production under normal operating conditions generates parts that consistently meet specifications over multiple runs or shifts. Sampling confirms key dimensions, weight, appearance, and process capability indices (Cpk). Completion of PQ, together with customer approval when applicable, authorizes full-scale production. Secondary operations and assembly may be included as part of validated post-processing.
Critical Parameters Monitored During Validation
Process Parameters
Temperature, pressure, timing, and screw position are continuously monitored. Melt and mold temperatures, injection and hold pressures, cooling times, and cycle durations must remain within tight tolerances to ensure uniform filling, packing, and mechanical integrity. Advanced micro injection molding or insert molding applications require even stricter control.
Material Parameters
Material quality is verified through pre-drying of hygroscopic polymers, lot traceability, certificates of analysis, and melt flow assessments where applicable. Common materials include TPE, TPU, PET, PP, ABS, Nylon, and LSR. Moisture content directly affects void formation, surface defects, and mechanical performance.
Output Parameters
Key outputs include part dimensions, weight, visual quality, and statistical process capability (Cpk). Industrial parts generally require Cpk ≥1.33, while medical and safety-critical parts require Cpk ≥1.67, ensuring the process reliably produces conforming parts without manual intervention.
Pre-Validation Work and Supporting Systems
FMEA is conducted before formal validation to identify potential failure modes, calculate risk priority numbers, and prioritize high-risk items for mitigation. Robust documentation and traceability systems include:
- Mold History Records (MHR): design drawings, modification logs, steel certificates, maintenance records, trial outcomes, and cumulative mold cycles
- Batch Production Records: actual process parameters, material lot numbers, inspection results
- Preventive Maintenance Logs
Operators must undergo certification, adhere to SOPs, and record all activities. High-performance molds with uniform wall thickness, polished surfaces, and conformal cooling help ensure dimensional stability and process repeatability.
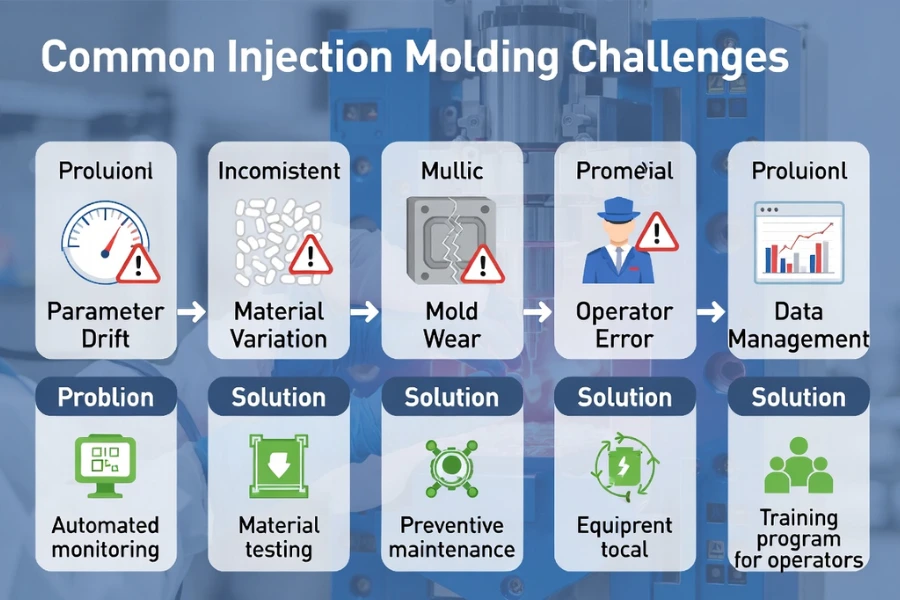
Common Challenges and Mitigation Strategies
| Challenge | Manifestation | Mitigation |
|---|---|---|
| Parameter drift | Temperature or pressure deviations over extended runs | Real-time monitoring, scheduled calibration, preventive maintenance |
| Material batch variation | Differences in flow behavior or moisture | Incoming material testing, supplier specifications, representative validation batches |
| Mold wear | High-performance polymers accelerate wear | Periodic inspection, component replacement, mold lifecycle documentation |
| Operator variability | Parameter reset errors or missing logs | Standardized SOPs, operator certification, automated mold change systems |
| Data management | Lost or inconsistent records | Electronic data collection, regular backups, audit trail functionality |
| Sterility requirements | Contamination from environment or personnel | Cleanroom molding (Class 7/8 cleanroom injection molding), robotic handling, reduced manual contact |
Post-Validation Monitoring and Revalidation
Daily monitoring using SPC charts tracks dimensions and weights to detect trends early. Revalidation is recommended periodically or when significant changes occur, such as:
- Mold relocation or major maintenance
- New material suppliers
- Equipment upgrades or modifications
- Production pauses exceeding 12 months
This ensures the process remains in a validated state over the lifecycle of production.
FAQ – Industry Common Questions
Q1: What is the main difference between OQ and PQ?
OQ defines operational limits and the process window, whereas PQ demonstrates that normal production consistently produces conforming parts with acceptable Cpk.
Q2: What Cpk values are required to pass validation?
General industrial parts: Cpk ≥1.33; medical or safety-critical parts: Cpk ≥1.67. Lower values require tighter tolerances or reduced variability.
Q3: Are full IQ/OQ/PQ phases required for small batches?
Yes, with PQ possibly using representative batches (3–5) combined with process justification.
Q4: What does a Mold History Record include?
MHR contains design drawings, modification records, steel certificates, maintenance logs, trial outcomes, and cumulative mold cycles.
Q5: Which biocompatibility tests must medical parts pass?
ISO 10993 tests include cytotoxicity, sensitization, irritation, systemic toxicity, hemocompatibility, and genotoxicity, depending on use and exposure.
Q6: What does FDA 21 CFR 820 audit in injection molding?
Audits focus on execution of IQ/OQ/PQ, complete batch/parameter records, CAPA system, and supplier compliance.
Q7: Are defective parts allowed during validation runs?
No, unless defects are clearly attributable to unusual causes like material contamination.
Q8: How can facilities without automation perform validation?
Manual recording is acceptable if schedules, personnel, and forms are strictly followed, with traceable signatures and dates.
Soft Marketing Integration
SeaSkyMedical provides ISO 13485-certified medical injection molding services, offering end-to-end solutions from mold design and prototyping to full-scale production in Class 7/8 cleanroom environments. By combining precision tooling, validated processes, and comprehensive documentation, SeaSkyMedical ensures clients consistently achieve compliant, high-quality medical components for complex and critical applications.




