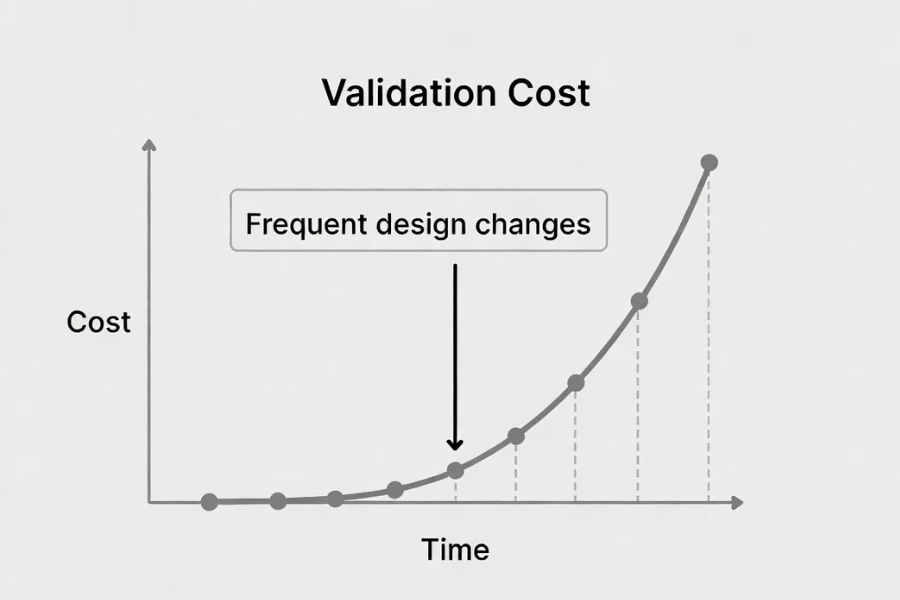
What Validation Cost Means in Medical Injection Molding
Validation cost in medical plastic injection molding refers to the total effort required to demonstrate that a manufacturing process consistently produces compliant parts under controlled and documented conditions, which typically includes installation verification, process characterization, performance confirmation, traceability setup, and regulatory documentation aligned with ISO 13485 expectations.
When design changes occur, this cost does not increase in a linear way, because each modification can invalidate previously qualified assumptions, meaning that earlier validation evidence may no longer be acceptable and must be partially or fully regenerated depending on the level of risk introduced by the change.
What Happens to Validation Cost When Design Changes Occur
Frequent design changes increase validation-related expenses because they disrupt the stability of the qualified process, and once stability is compromised, manufacturers are required to re-establish process capability, confirm repeatability, and update documentation to maintain compliance.
In practical terms, a geometry adjustment that alters flow behavior or stress distribution can shift the established process window, which then requires additional experimentation and verification work, often supported by early-stage mold design and DFM review, while a material change or tolerance shift can expand the scope further by introducing new regulatory and performance considerations.
This is why, in most controlled medical manufacturing environments, design modifications are treated as validation triggers rather than simple engineering updates, since their impact extends beyond tooling or production into compliance, audit readiness, and long-term product reliability.
Core Relationship Between Design Changes and Validation Cost Growth
In medical molding programs, design change frequency and validation cost are strongly correlated because each change introduces uncertainty into a system that has already been qualified for stability, and restoring that stability requires additional testing, documentation, and controlled production evidence.
The effect becomes more pronounced when hardened production tooling is involved, particularly in medical mold making, since tooling modifications are not only expensive in themselves but also tied to process requalification, which means that a single change can cascade into multiple validation activities across different stages of the manufacturing lifecycle.
From an engineering perspective, this relationship can be understood as a shift from manufacturing cost to qualification cost, where the dominant expense driver becomes the effort required to prove that the process remains valid under the updated conditions rather than the cost of producing the part itself.
How Design Changes Propagate Through Validation Stages
Impact on Installation Qualification
When a design change alters the physical structure of the mold, such as modifying gate location, venting strategy, cavity geometry, or introducing new side-action mechanisms, the original installation assumptions are no longer fully applicable, because the tooling configuration has changed and must be reverified against defined requirements.
This typically leads to partial or extended installation verification, especially if the change affects auxiliary systems, fixturing, or cleanroom handling setups, where additional checks are required to confirm that the updated configuration performs consistently within controlled environments such as cleanroom injection molding.
From a process standpoint, this stage remains relatively moderate in cost, but it establishes the foundation for downstream validation, which means that incomplete verification at this level can amplify issues later in the qualification process.
Impact on Operational Qualification
Operational qualification is particularly sensitive to design changes because it depends on a stable relationship between material behavior, mold geometry, and processing conditions, and when one of these variables shifts, the previously defined process window may no longer produce consistent results.
For example, a change in wall thickness or internal geometry can alter flow front behavior, which affects weld line formation, cooling gradients, and shrinkage distribution, ultimately leading to dimensional variation that must be re-evaluated through controlled experimentation.
This requirement to re-run process development activities is a primary contributor to increased process validation cost, as it involves multiple production runs, machine time, and data analysis before a new stable operating range can be confirmed.
Impact on Performance Qualification
Performance qualification represents the stage where process consistency is demonstrated under routine production conditions, and because it relies on repeatability across consecutive batches, any design change that affects process stability can require this stage to be repeated.
The impact becomes more significant when the change influences critical-to-quality characteristics or sterilization performance, as additional testing such as aging studies may be required to confirm long-term stability.
As a result, this stage often becomes the dominant cost driver after design modifications, since it combines production effort, testing, and documentation into a single validation activity required for product release.
How Tooling Strategy Amplifies or Mitigates Change-Driven Cost
The cost impact of design changes is closely tied to the type of tooling and manufacturing approach used, because different methods offer varying levels of flexibility and validation dependency.
| Tooling Approach | Change Flexibility | Validation Impact | Typical Use Stage |
|---|---|---|---|
| Additive or Casting Methods | High | Minimal validation dependency | Early concept and iteration |
| CNC Machining | High | Limited to dimensional verification | Pre-validation development |
| Bridge Tooling | Moderate | Partial requalification | Late development and clinical builds |
| Hardened Production Tooling | Low | Full or near-full requalification | Post design freeze production |
Bridge tooling is often used to balance flexibility and validation effort, as it allows controlled iteration while limiting the scope of requalification, whereas hardened steel tooling tends to lock both geometry and process conditions, making any subsequent modification significantly more expensive.
In practice, this evaluation is typically part of a pre-tooling feasibility review, where experienced partners such as SeaSkyMedical assess tooling strategy alongside product development services to align design maturity with validation requirements.
High-Risk Design Change Scenarios That Drive Validation Cost
Material Changes and Regulatory Impact
Material changes introduce one of the highest levels of validation impact because they directly affect biocompatibility, sterilization compatibility, and processing characteristics, all of which must be supported by documented evidence.
A change in resin grade or supplier can alter chemical composition and performance behavior, requiring renewed biological evaluation, sterilization validation, and process requalification, which significantly increases both cost and timeline.
Geometry Changes That Affect Flow and Stress Distribution
Modifications involving undercuts, thin walls, or complex internal features influence how the polymer fills and solidifies, which affects dimensional stability and structural integrity.
When these changes require advanced tooling mechanisms, including solutions such as 2K injection molding, the validation scope expands further due to additional mechanical and process considerations.
Changes to Critical-to-Quality Dimensions
Adjustments to dimensions that define product function require reassessment of process capability, since even small deviations can impact performance or compliance outcomes.
This typically involves repeating capability studies and confirming that the process can consistently meet specification limits.
Regulatory and Documentation Burden of Frequent Design Changes
In medical manufacturing, design changes extend into documentation and regulatory processes, where each modification must be recorded, justified, and integrated into the design history file and risk management documentation.
Depending on the extent of the change, regulatory submissions or notifications may be required, which introduces additional cost and administrative effort.
From an audit perspective, frequent changes without structured control increase the risk of nonconformities, as quality systems require traceability and defined validation logic for all modifications.
Strategies to Control Validation Cost Under Frequent Design Iteration
Use Flexible Manufacturing Methods Before Design Freeze
During early development stages, when design changes are expected, manufacturing methods such as CNC plastic machining or 3D prototype printing allow rapid iteration without triggering repeated qualification cycles.
As the design stabilizes, transitioning to bridge tooling provides a controlled pathway toward production readiness while maintaining flexibility for minor adjustments.
Apply Modular Tooling Concepts
Designing molds with interchangeable inserts allows localized changes without modifying the entire tool, which helps limit both tooling cost and requalification scope.
Establish Risk-Based Validation Planning
A structured validation plan that defines how different types of changes are handled allows manufacturers to avoid unnecessary requalification while maintaining compliance with quality requirements.
Use Process Monitoring to Support Reduced Requalification
When process parameters remain within established control limits, statistical evidence can support reduced validation scope if this approach is defined within the quality framework.
Integrate Early DFM and Feasibility Review
Early design for manufacturability review, supported by product development services, helps identify validation-sensitive features before tooling is finalized.
Working with SeaSkyMedical allows design teams to evaluate tooling feasibility and validation implications together, improving decision-making before production commitment.
When Injection Molding Becomes a High-Risk Choice
Injection molding is not always the optimal method when design stability has not been achieved, particularly when production volume is low or design changes remain frequent.
In such cases, alternative manufacturing approaches provide greater flexibility and reduce validation-related expenses.
FAQ
When does a design change require re-validation
A design change typically requires re-validation when it affects process stability, material behavior, or critical product characteristics, as these factors determine whether existing validation evidence remains applicable.
How should validation cost be estimated before approving a design change
Validation cost estimation should include tooling modification, process requalification, testing, and documentation updates, as these collectively define the total impact of a change.
Can validation scope be reduced after minor design adjustments
Validation scope can sometimes be reduced if the change has no measurable impact on process capability or product performance, but this requires predefined criteria and supporting data.
Why do material changes have a larger impact than geometry changes
Material changes affect both processing behavior and regulatory compliance, which means they typically require broader evaluation compared to geometry-only modifications.
Conclusion
Frequent design changes in medical injection molding programs should be understood as events that disrupt validated process conditions, requiring additional effort to restore compliance and stability through structured qualification activities.
By aligning design decisions with validation strategy and integrating early engineering review, manufacturers can reduce unnecessary requalification cycles and manage cost more effectively.
SeaSkyMedical supports medical device contract manufacturing programs through feasibility evaluation, tooling strategy planning, and validation-oriented manufacturing support, helping teams move toward stable production with greater confidence.