Medical injection molded components must comply with strict regulatory frameworks. These requirements ensure patient safety, product reliability, and consistent manufacturing quality.
Plastic components used in healthcare applications are regulated through international standards and national regulatory systems. The most common frameworks include ISO quality standards, FDA regulations, biocompatibility requirements, and controlled manufacturing environments.
This article explains the regulatory standards medical injection molded parts must meet and how these standards influence manufacturing processes.
What Are Medical Injection Molded Parts
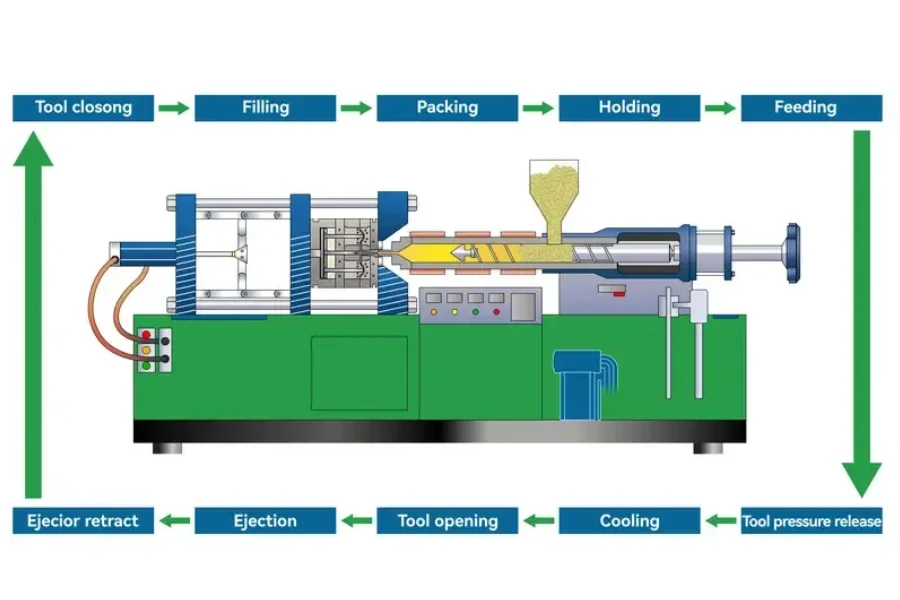
Definition of Medical Injection Molded Components
Medical injection molded parts are plastic components manufactured through injection molding for use in medical devices and healthcare equipment.
Molten medical-grade polymers are injected into precision molds and cooled to create components with controlled geometry and repeatable dimensions. This process allows manufacturers to produce large quantities of parts with consistent quality.
Injection molding is commonly used when medical devices require tight tolerances, stable material performance, and repeatable production conditions.
Typical Examples of Medical Injection Molded Parts
Medical injection molding supports a wide range of healthcare components.
Typical examples include
- syringes
- IV connectors
- catheter hubs
- diagnostic cartridges
- Petri dishes
- medical device housings
Many of these products are manufactured in high volumes where consistent dimensions and material behavior are critical.
Why Regulatory Compliance Is Critical in Medical Manufacturing
Medical products operate in environments where component failure may directly affect patient safety.
For this reason, regulatory systems require manufacturers to control several aspects of production
- material safety
- contamination risks
- process consistency
- traceability of production batches
- documentation of manufacturing parameters
Even small molded plastic components can affect the performance of a medical device. Regulatory frameworks help ensure that products perform consistently and safely across all production batches.
Core Regulatory Standards for Medical Injection Molded Parts
Medical injection molded components typically need to comply with multiple international standards and regulatory frameworks.
These standards define how products are designed, manufactured, validated, and documented throughout their lifecycle.
ISO 13485 Quality Management System for Medical Devices
ISO 13485 defines the quality management requirements for organizations involved in medical device manufacturing.
The standard establishes a framework for controlling manufacturing processes and maintaining traceability across production.
Key areas addressed by ISO 13485 include
- design and development control
- risk management procedures
- supplier qualification
- document and record management
- product traceability and batch control
Manufacturers operating under ISO 13485 must maintain detailed production documentation, including Device Master Records and Device History Records. These records allow manufacturers and regulators to trace every production batch back to the raw materials, equipment, and operators involved in production.
ISO 10993 Biological Evaluation of Medical Devices
ISO 10993 focuses on evaluating the biological safety of materials used in medical devices.
The standard defines testing methods used to assess whether a material may cause harmful biological reactions when exposed to the human body.
Typical evaluations include
- cytotoxicity testing
- sensitization testing
- irritation testing
- implantation response testing
The required tests depend on the duration and type of contact between the device and the human body. This evaluation is often performed during material selection and device design.
ISO 14644 Cleanroom Manufacturing Requirements
Many medical injection molded components are produced in controlled environments known as cleanrooms.
ISO 14644 defines the classification and performance requirements for these environments. The standard regulates factors such as airborne particle concentration and environmental monitoring procedures.
Medical injection molding operations commonly use ISO Class 7 or ISO Class 8 cleanrooms to reduce contamination risks during production.
Cleanroom environments control airborne particles, temperature, and humidity to prevent contamination of sensitive medical components.
FDA Regulatory Requirements for Medical Devices
In the United States, medical device manufacturing must comply with FDA Quality System Regulation defined in 21 CFR Part 820.
These regulations require manufacturers to establish documented procedures covering the entire product lifecycle.
Key regulatory elements include
- design controls
- process validation
- corrective and preventive action systems
- complaint handling procedures
- device history record documentation
FDA inspections evaluate whether manufacturers follow these documented procedures and maintain adequate production records.
For injection molding processes, this typically requires validation of process parameters and ongoing monitoring of manufacturing consistency.
Medical Device Classification and Its Impact on Manufacturing Requirements
Medical devices are classified according to their potential risk to patients.
The classification level determines the amount of regulatory oversight required.
Class I Devices
Class I devices represent the lowest risk category.
Examples include
- laboratory containers
- hospital equipment
- test tubes
These products generally require basic regulatory controls.
Class II Devices
Class II devices represent moderate risk.
Examples include
- syringes
- infusion pumps
- diagnostic equipment
These devices typically require additional performance testing and regulatory documentation.
Class III Devices
Class III devices represent the highest risk category.
Examples include
- pacemakers
- implantable prosthetic devices
These products require extensive clinical evaluation and strict manufacturing control.
For injection molded components used in higher risk devices, manufacturers must maintain stricter validation and traceability procedures.
Injection Molding Process Validation Under FDA Requirements
Medical injection molding processes must be validated to demonstrate consistent production performance.
Validation confirms that manufacturing processes can repeatedly produce parts that meet quality specifications.
Installation Qualification IQ
Installation Qualification verifies that molding equipment and tooling are installed correctly.
This step confirms that machines, molds, and supporting systems meet design and operational requirements.
Operational Qualification OQ
Operational Qualification verifies that the molding process operates within defined parameter ranges.
Typical parameters evaluated include
- injection pressure
- melt temperature
- cooling time
- mold temperature
- cycle time
These parameters are tested to identify acceptable operating ranges for stable production.
Performance Qualification PQ
Performance Qualification confirms that the validated process consistently produces acceptable parts during routine production.
Production samples are evaluated for
- dimensional accuracy
- surface quality
- mechanical performance
PQ demonstrates that the injection molding process remains stable during continuous manufacturing.
Material Compliance Requirements for Medical Injection Molding
Materials used in medical injection molding must meet strict safety and performance requirements.
Medical grade plastics are typically produced using virgin materials rather than recycled polymers to reduce contamination risks.
Key material requirements include
- biocompatibility
- resistance to sterilization processes
- chemical stability
- mechanical strength
Common medical polymers include
| Material | Typical Medical Applications |
|---|---|
| Polypropylene PP | syringes and containers |
| Polyethylene PE | tubing and packaging |
| Polycarbonate PC | diagnostic device housings |
| PEEK | implantable device components |
| Medical silicone | catheters and flexible seals |
Material selection must also consider sterilization compatibility and regulatory approval status.
Sterilization Compatibility Requirements
Many medical components must withstand sterilization before clinical use.
Injection molded parts must maintain structural integrity after exposure to sterilization processes.
Common sterilization methods include
- steam autoclaving
- gamma radiation
- ethylene oxide sterilization
Some polymers may degrade under repeated sterilization cycles. For this reason material compatibility testing is typically performed during device development.

Cleanroom Manufacturing and Contamination Control
Contamination control is a key requirement in medical device manufacturing.
Cleanroom environments regulate several factors
- airborne particle concentration
- microbial contamination
- environmental conditions
Environmental monitoring systems track particle levels and ensure that manufacturing conditions remain within acceptable limits.
Manufacturers specializing in medical injection molding often operate ISO Class 7 or ISO Class 8 cleanroom facilities to reduce contamination risks during production.
Facilities such as SeaSkyMedical operate controlled manufacturing environments designed for medical plastic component production combining precision molding processes with regulated cleanroom conditions.
Applications of Medical Injection Molded Components
Medical injection molding supports a wide range of healthcare products.
Typical applications include
- disposable medical consumables
- diagnostic laboratory equipment
- catheter connectors and hubs
- surgical instrument components
- device housings and enclosures
Injection molding allows manufacturers to produce these components with precise dimensions and stable material properties across large production volumes.
Conclusion
Medical injection molded components must comply with multiple regulatory frameworks to ensure safety and reliability.
Important regulatory systems include
- ISO 13485 quality management systems
- ISO 10993 biological safety evaluation
- ISO 14644 cleanroom manufacturing standards
- FDA Quality System Regulation under 21 CFR Part 820
These standards control how medical components are designed manufactured validated and documented.
Manufacturers specializing in medical plastic molding operate controlled quality systems and regulated production environments to ensure that injection molded components meet these regulatory requirements.
FAQ
What ISO standards apply to medical injection molding
Medical injection molding commonly follows ISO 13485 for quality management ISO 10993 for biological safety evaluation and ISO 14644 for cleanroom manufacturing environments.
What FDA regulation governs medical device manufacturing
Medical device manufacturing in the United States follows FDA Quality System Regulation defined in 21 CFR Part 820.
Why are cleanrooms required for medical injection molding
Cleanrooms control airborne particles and contamination during production. This helps protect sensitive medical components and maintain regulatory compliance.
What is IQ OQ PQ validation
IQ OQ PQ validation is a structured process used to confirm that manufacturing equipment and processes consistently produce products that meet quality specifications.
What materials are commonly used for medical injection molded parts
Common materials include polypropylene polyethylene polycarbonate PEEK and medical grade silicone used in medical devices and laboratory components.




