Medical devices and laboratory instruments must remain sterile during clinical use. Many reusable tools are exposed to sterilization cycles repeatedly during their service life.
These cycles expose materials to heat, pressure, moisture, chemicals, or radiation. Selecting polymers that maintain mechanical integrity and dimensional stability under these conditions is an important part of medical product development.
Why Repeated Sterilization Matters in Medical Device Design
Sterilization removes microorganisms from instruments that come into contact with patients or biological samples. Reusable medical tools often undergo hundreds of sterilization cycles during their operational lifetime.
Repeated exposure to heat and moisture can gradually degrade many polymers. Materials that perform well in standard environments may lose strength, warp, or become brittle after multiple sterilization cycles.
For this reason, engineers typically evaluate medical plastic material selection early in the product development process.
Common Sterilization Methods Used in Healthcare
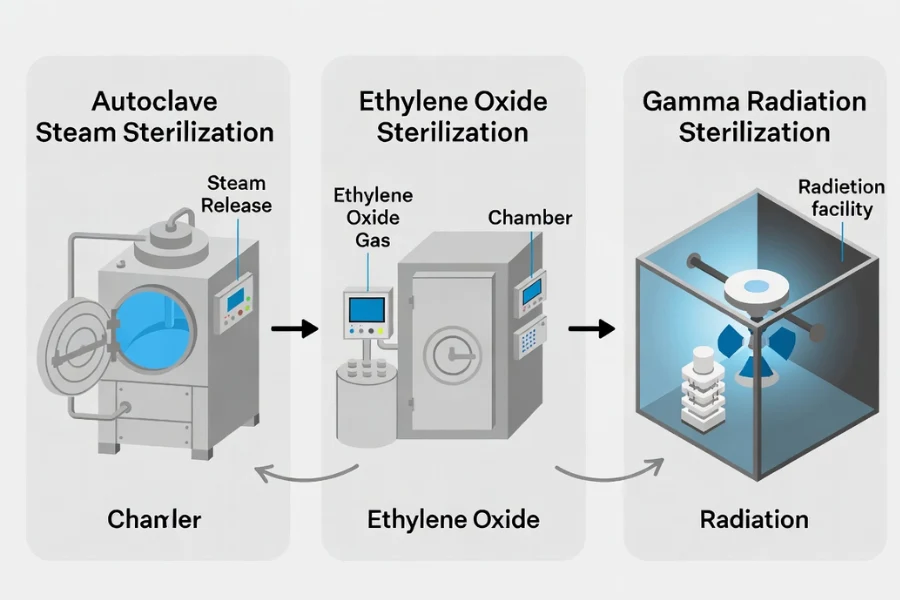
Different sterilization methods expose materials to different environmental stresses.
Steam Sterilization (Autoclave)
Steam sterilization is widely used in hospitals and laboratories.
Typical conditions include:
- Temperatures between 121–134°C
- Pressurized saturated steam
- Repeated heating and cooling cycles
Steam sterilization is highly effective but places significant thermal and hydrolytic stress on many plastics.
Ethylene Oxide Sterilization
Ethylene oxide sterilization is commonly used for temperature-sensitive devices.
Characteristics include:
- Low temperature processing
- Gas penetration into complex assemblies
- Extended aeration to remove residual gas
Many polymers tolerate this method well, although some materials may absorb residual gas.
Gamma Radiation Sterilization
Gamma radiation sterilization uses high-energy photons to disrupt microbial DNA.
Typical features include:
- No moisture exposure
- Sterilization after final packaging
- Industrial scale processing
However, radiation may alter polymer chains, which can lead to brittleness or discoloration in some plastics.
Hydrogen Peroxide Plasma Sterilization
This method operates at lower temperatures and is suitable for heat-sensitive medical equipment.
Reactive plasma environments can oxidize certain polymers, so compatibility testing is necessary.
Why Many Plastics Fail Under Repeated Sterilization
Many general-purpose plastics degrade when exposed to sterilization conditions.
Common failure mechanisms include:
- Hydrolysis caused by hot moisture
- Thermal deformation when temperatures approach the glass transition temperature
- Stress cracking during repeated heating and cooling cycles
- Material embrittlement caused by polymer chain degradation
- Surface discoloration from radiation exposure
These effects accumulate gradually across repeated sterilization cycles.
Key Material Properties Required for Repeated Sterilization

Materials used in sterilizable medical components must maintain their properties after multiple sterilization cycles.
Key characteristics include:
- High heat resistance above sterilization temperatures
- Strong hydrolysis resistance during steam exposure
- Dimensional stability under thermal cycling
- Mechanical durability without brittleness
- Chemical resistance against sterilizing agents
- Biocompatibility for medical applications
Medical-grade polymers used in patient-contact applications are often evaluated under frameworks such as ISO 10993 for biological safety.
Common Plastics Suitable for Repeated Sterilization

Several high-performance polymers are widely used for medical components that require repeated sterilization.
| Material | Typical Sterilization Durability | Key Characteristics | Typical Medical Uses |
|---|---|---|---|
| PEEK | 1000+ cycles | Exceptional thermal stability and chemical resistance | Surgical tools, orthopedic guides |
| PPSU | 1000+ cycles | High impact strength and hydrolysis resistance | Sterilization trays, reusable housings |
| PEI (Ultem) | 500–2000 cycles | Rigid, high glass transition temperature | Medical equipment enclosures |
| PTFE | Very high | Chemically inert and thermally stable | Tubing liners, seals |
| Silicone | High | Flexible and heat resistant elastomer | Medical tubing, seals |
| Polypropylene (PP) | 100–200 cycles | Moderate heat resistance and cost efficiency | Laboratory containers, syringe components |
Actual lifespan depends on part geometry, sterilization parameters, and mechanical loading during use. Components such as lab containers and syringe elements are often produced using polypropylene injection molding.
Materials That Typically Do Not Perform Well in Steam Sterilization
Some common plastics cannot tolerate repeated steam sterilization.
| Material | Typical Issue |
|---|---|
| Polycarbonate | Hydrolysis leads to embrittlement and loss of transparency |
| PVC | Softening and additive migration under heat |
| Polystyrene | Deformation at relatively low temperatures |
| PET or PETG | Dimensional instability during steam exposure |
| Standard polyethylene | Low melting temperature |
These materials may still be suitable for single-use devices or alternative sterilization methods such as gamma radiation.
Material Selection Considerations for Injection Molded Medical Parts
Material selection should be evaluated alongside manufacturing requirements. Many reusable medical components are produced through medical plastic injection molding to achieve consistent quality and tight dimensional control.
Injection molded medical components must retain both dimensional accuracy and mechanical strength after sterilization cycles.
Important considerations include:
- Expected number of sterilization cycles during the product lifecycle
- Mechanical loads applied during clinical use
- Wall thickness distribution and part geometry
- Compatibility with sterilization environments
Sterilizable medical components are often manufactured in controlled environments such as cleanroom injection molding facilities to minimize contamination during production.
Small and high-precision medical parts may also require micro injection molding processes to maintain tight tolerances.
SeaSkyMedical works with medical device developers to support material selection, mold design, and precision molding of sterilization-resistant plastic components.
Design Considerations for Sterilizable Injection Molded Parts
Part design can significantly influence sterilization durability.
Recommended design practices include:
- Maintaining uniform wall thickness to reduce thermal stress
- Avoiding sharp internal corners that concentrate stress
- Designing geometries that prevent moisture accumulation
- Selecting polymers compatible with the intended sterilization method
Long-term performance also depends on proper tooling and medical mold making. Early-stage mold design services can help engineers ensure manufacturability and stable production.
Typical Medical Applications Requiring Repeated Sterilization
Repeated sterilization is common in reusable medical equipment.
Typical applications include:
- Surgical instrument components
- Sterilization trays and instrument holders
- Reusable diagnostic device housings
- Laboratory testing equipment
- Medical device handling fixtures
Many of these products are manufactured as custom medical components for diagnostic, surgical, and laboratory systems.
FAQ
Which plastics can withstand repeated autoclave sterilization
High-performance polymers such as PEEK, PPSU, PEI, and PTFE are commonly used for components that undergo repeated steam sterilization. These materials maintain mechanical integrity and dimensional stability during multiple sterilization cycles.
Is polypropylene suitable for repeated sterilization
Polypropylene can tolerate limited steam sterilization cycles depending on material grade and design. It is often used for laboratory containers and syringe components that experience moderate sterilization exposure.
Why does polycarbonate fail during repeated autoclave cycles
Polycarbonate is susceptible to hydrolysis when exposed to hot moisture. Over multiple steam sterilization cycles, the polymer chains degrade, causing embrittlement and loss of transparency.
What factors influence sterilization compatibility of plastic parts
Material composition, sterilization temperature, moisture exposure, part geometry, and residual molding stress all influence how a plastic component performs during repeated sterilization cycles.
Can injection molded medical parts be designed for sterilization durability
Yes. Proper material selection, uniform wall thickness, and controlled molding processes help ensure that injection molded medical parts maintain structural integrity during repeated sterilization.
Conclusion
Repeated sterilization exposes medical components to challenging environments involving heat, pressure, moisture, chemicals, or radiation. Only a limited group of high-performance polymers can maintain their mechanical strength and dimensional stability under these conditions.
Materials such as PEEK, PPSU, PEI, PTFE, silicone, and certain grades of polypropylene are commonly used in reusable medical devices because they tolerate repeated sterilization cycles.
Careful coordination between material selection, product design, and manufacturing processes helps ensure long-term reliability. SeaSkyMedical provides medical device contract manufacturing services that support the production of sterilization-compatible plastic components used in medical and laboratory applications.