Why Process Data Recording Sits at the Center of Regulatory Audits
Manufacturers record process data for regulatory audits by combining automated data capture, validated digital systems, and audit trail mechanisms that ensure every production step is time-stamped, traceable, and protected from unauthorized modification, allowing auditors to reconstruct the full manufacturing history without relying on interpretation.
In regulated manufacturing environments such as pharmaceuticals, medical devices, food processing, and automotive production, regulatory audits are not limited to verifying finished product quality, as they are designed to confirm that each process step has been executed under controlled and documented conditions.
Regulatory bodies such as the FDA and EMA require manufacturers to demonstrate not only that products meet specifications, but that every production stage is supported by complete, transparent, and tamper-resistant records that remain consistent throughout the data lifecycle.
Without a structured data recording system, manufacturers face measurable risks including warning letters, production disruption, product recalls, and long-term damage to operational credibility.
What Process Data Recording and Audit Trails Actually Mean
Process Data Recording in Manufacturing
Process data recording refers to the systematic capture and storage of all critical manufacturing information across the production lifecycle, including raw material identification, machine parameters, environmental conditions, inspection results, operator actions, and time-based events, all of which must remain consistent, attributable, and audit-ready.
In practice, this means that data is captured either automatically through connected equipment or through controlled system input, with each entry linked to a specific batch, process step, and operator identity.
Audit Trails as the Backbone of Data Integrity
An audit trail is a secure, computer-generated, time-stamped record that logs every action performed within a system, ensuring that no data can be altered without leaving a permanent and reviewable trace.
A compliant audit trail captures who performed the action, when it occurred, where it was executed, what was changed, why the change was made when required, and how the process was carried out, forming a complete reconstruction path for auditors.
Unlike simple logs or spreadsheets, audit trails must be complete, automatically generated, and designed so that original data is never overwritten, with any modification recorded as a new entry linked to the original value.
Regulatory Frameworks That Govern Process Data Recording
Manufacturers record process data in a structured and controlled way because regulatory frameworks explicitly require that electronic records remain reliable, traceable, and protected throughout their lifecycle.
FDA 21 CFR Part 11 requires electronic records and signatures to be trustworthy and equivalent to paper records, supported by audit trails, system validation, and controlled access.
EU GMP Annex 11 imposes similar requirements within the European Union, with a strong emphasis on data integrity and the secure operation of computerized systems used in production and quality processes.
ALCOA+ principles define the baseline expectations for data, requiring that it is attributable, legible, contemporaneously recorded, original, and accurate, while also being complete, consistent, enduring, and available over time.
Industry-specific standards such as GMP, HACCP, IATF 16949, and ISO 13485 extend these requirements into sector-specific expectations, but consistently require that audit trails accompany records throughout their lifecycle and are subject to periodic review.
How Process Data Is Recorded Across a Typical Manufacturing Workflow
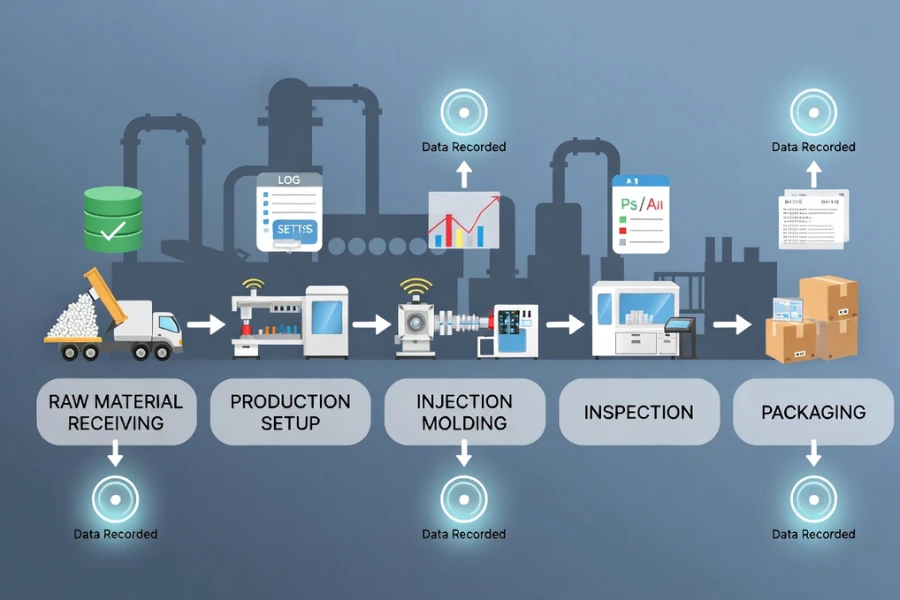
To understand how process data is recorded in practice, it is necessary to follow a product through a real manufacturing workflow, where each stage generates specific data that must be captured, validated, and linked to an audit trail.
Raw Material Receiving
When materials enter the facility, batch numbers, supplier information, certificates of analysis, and receiving inspection results are recorded through ERP systems or barcode-based identification, ensuring that each material lot can be traced back to its origin and linked to downstream production batches.
Production Setup
During setup, machine configuration parameters, mold identification, calibration status, and operator login credentials are recorded through MES systems, where each setup action is time-stamped and associated with a specific production order, ensuring that the initial process state is fully documented.
Accurate process control at this stage is closely tied to tooling quality, and well-defined mold making directly influences parameter stability and repeatability throughout production.
Processing Stage
In processes such as medical plastic injection molding, where parameters must be tightly controlled and continuously recorded, key variables including melt temperature, injection pressure, holding pressure, cooling time, and cycle time are typically captured at high frequency through machine controllers or integrated systems.
For example, melt temperatures may be recorded continuously within defined ranges, injection pressure curves may be captured per cycle, and cycle times are automatically logged to detect deviations from validated process windows, while operator interventions such as parameter adjustments are recorded as audit trail events rather than overwriting previous values.
In regulated environments such as cleanroom injection molding, environmental data including particulate levels, humidity, and temperature must also be continuously monitored and recorded to ensure compliance with controlled manufacturing conditions.
For applications requiring extremely tight tolerances, such as micro injection molding, process data recording becomes even more critical, as small parameter deviations can directly impact product performance.
Complex processes such as medical insert molding require additional layers of data capture, including insert positioning and multi-step validation records.

Inspection and Quality Control
Inspection results, measurement data, and deviation records are captured through LIMS or MES quality modules, where each result is linked to a batch, operator, and timestamp, ensuring that quality decisions can be traced back to both process conditions and measurement outcomes.
Packaging and Release
At the final stage, packaging verification, labeling checks, and release approvals are documented through electronic batch records, where electronic signatures confirm that all required process steps and quality checks have been completed before product release.
Downstream operations such as medical device assembly and medical device packaging also require controlled data recording to ensure traceability beyond primary manufacturing.
Across this workflow, all data entries, approvals, and changes are automatically linked to audit trails, ensuring that original records remain intact and any modification is fully documented.
Core Technologies Used to Record Process Data
Manufacturers rely on a combination of automated data acquisition and integrated digital systems to ensure that process data is recorded accurately and consistently across all production stages.
Automated data acquisition through sensors, barcode systems, RFID tags, and connected equipment allows process parameters to be captured in real time, reducing manual input and minimizing human error while supporting contemporaneous recording requirements.
MES platforms serve as the central layer for capturing production data, linking machine inputs, operator actions, and quality checks into structured records, while LIMS systems manage laboratory data and ensure that analytical results are recorded with full traceability and audit trails.
ERP systems provide supply chain traceability, and data historians or cloud-based platforms store large volumes of time-series data for long-term retention and rapid retrieval.
Electronic batch records replace paper-based documentation by capturing every production step digitally, automatically generating audit trails for approvals, parameter changes, and deviations.
Supporting processes such as 3D prototype printing and CNC plastic machining also benefit from structured data recording, particularly during validation and pre-production stages.
Integrated capabilities such as custom plastic molding further support consistent data capture across different product types and manufacturing requirements.
Best Practices for Ensuring Data Integrity
Ensuring data integrity requires validated systems, controlled access, and continuous oversight to maintain the reliability of recorded data throughout its lifecycle.
All systems used in regulated activities must be validated, while role-based access control ensures that only authorized personnel can create, modify, or approve records.
Audit trails must be reviewed at defined intervals based on process risk, ensuring that anomalies such as unexpected parameter changes or irregular timestamps are identified and addressed.
Automated backup systems protect data from loss, and long-term retention strategies ensure that records remain accessible throughout their required lifecycle.
Employee training ensures that personnel understand both system operation and data integrity principles, reducing the likelihood of errors and ensuring compliance.
How Traceability Audits Are Conducted in Practice
Traceability audits evaluate whether a manufacturer can reconstruct the complete history of a product using recorded data and audit trails, covering internal processes, supplier inputs, and distribution outputs.
Before an audit, manufacturers organize batch records, calibration data, inspection reports, and supplier documentation while conducting internal checks to confirm completeness.
During the audit, auditors typically select a batch and request a full reconstruction of its history, verifying that audit trails capture all changes and user actions without gaps.
Auditors may also simulate recall scenarios, requiring rapid identification of affected batches.
After the audit, corrective actions are implemented to address gaps and improve system performance.
Common Challenges and Practical Solutions
Manufacturers often face challenges related to system cost, legacy equipment integration, supplier data inconsistency, and increasing data storage requirements.
These challenges are typically addressed through phased system implementation, integration of legacy equipment via data gateways, standardized supplier requirements, and scalable storage strategies.
How Industry 4.0 Technologies Improve Data Recording
Advanced technologies enable real-time monitoring, predictive analysis, and improved data integrity across manufacturing operations.
AI identifies abnormal trends, cloud platforms centralize data access, and blockchain provides tamper-resistant audit trails, while digital twins allow validation of process and data recording logic before production begins.
Audit Readiness Checklist for Process Data Recording
Manufacturers can assess audit readiness by confirming that process data is automatically captured, that every record includes a complete audit trail, that systems prevent unauthorized modification, and that audit trail reviews are defined and documented.
They should also verify that user access is controlled and that data can be retrieved quickly during audits, as delayed or incomplete traceability is a common audit failure point.
If these questions cannot be answered immediately, the system may not be fully audit-ready.
Turning Data Recording Into a Competitive Advantage
Process data recording and audit trails form the foundation for operational control, quality consistency, and risk reduction in regulated manufacturing environments.
In medical manufacturing scenarios such as cleanroom production and precision injection molding, where parameters must remain tightly controlled and documentation must align with ISO 13485 expectations, having validated and integrated systems enables consistent compliance and production stability.
SeaSkyMedical supports this approach through integrated capabilities in medical device contract manufacturing, where process data recording, traceability, and quality control are aligned across injection molding, assembly, and cleanroom production environments.
Organizations that treat process data as a structured asset rather than a compliance burden are better positioned to meet regulatory requirements while improving efficiency and long-term reliability.
FAQ
Q1: How can small manufacturers meet audit trail requirements with limited budgets
Small manufacturers can prioritize cloud-based MES or LIMS systems while implementing electronic records in critical processes first and maintaining controlled paper systems where necessary.
Q2: What should be done if an audit trail shows unauthorized changes
An investigation should identify the user, timestamp, and reason for the change, followed by corrective action and root cause analysis.
Q3: Are paper records still acceptable for regulatory audits
Paper records are acceptable if they meet data integrity requirements, but electronic systems are increasingly preferred.
Q4: How can manufacturers prove audit trails are tamper-resistant
This is demonstrated through system validation, secure database design, restricted access, and protected storage mechanisms.
Q5: How long must audit trail data be retained
Audit trails must be retained for the same duration as associated records, often exceeding ten years depending on regulations.
Q6: Will auditors request recall simulations
Auditors often require manufacturers to trace a selected batch across the full production lifecycle within a defined timeframe to verify system effectiveness.




