ISO 13485 certification influences supplier selection by redefining the evaluation threshold. It transforms supplier approval from a commercial decision into a documented risk control decision.
Under ISO 13485, outsourced processes such as medical device contract manufacturing remain the responsibility of the medical device manufacturer. Certification therefore affects compliance posture, supplier onboarding cost, audit burden, and long-term regulatory exposure.
What ISO 13485 Requires for Supplier Control
ISO 13485 requires organizations to establish documented procedures to ensure purchased products conform to specified requirements.
Supplier control must include:
- Defined evaluation criteria
- Risk assessment tied to device impact
- Ongoing performance monitoring
- Documented verification activities
- Re-evaluation based on performance data
The standard does not allow risk transfer to suppliers. Even when production is outsourced, accountability remains with the device manufacturer.
This shifts supplier selection toward documented predictability rather than price competitiveness alone.
How ISO 13485 Changes the Supplier Evaluation Threshold
In practice, ISO 13485 introduces three structural changes to supplier selection.
1. Risk Tiering Becomes Mandatory
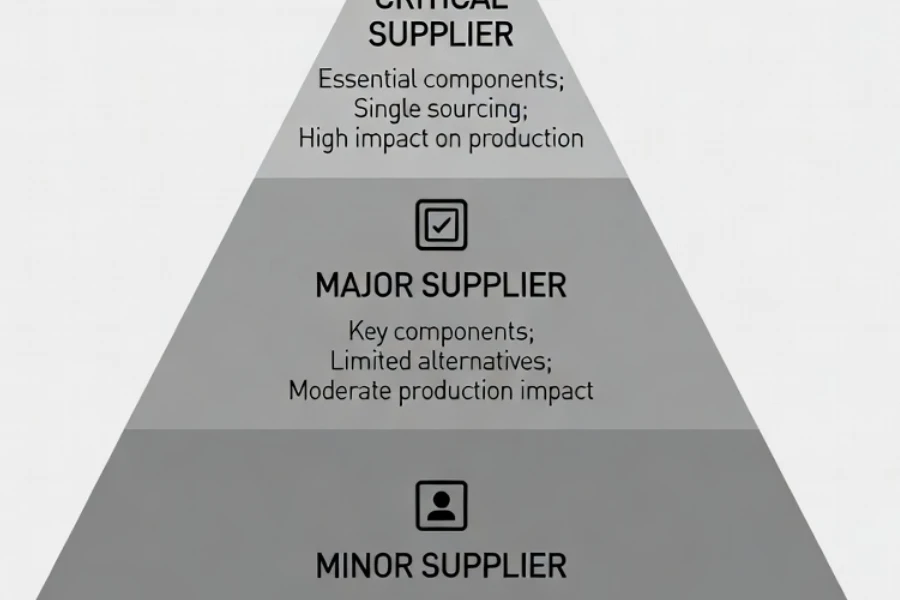
Suppliers are often classified into categories such as:
- Critical suppliers
- Major suppliers
- Minor suppliers
Critical suppliers may include those providing implantable components, sterile packaging, validated molding processes, or complex assembly operations.
Certification reduces uncertainty in risk classification because it demonstrates that a supplier already operates within a regulated quality framework.
2. Qualification Cost Becomes a Decision Variable
Supplier qualification requires:
- On-site audits
- Process validation review
- Documentation assessment
- Corrective action follow-up
A certified supplier typically requires fewer foundational audits because:
- Document control systems are established
- Change control procedures are defined
- Internal audits are already conducted
This reduces onboarding friction and accelerates supplier approval timelines.
3. Audit Burden Influences Procurement Decisions
Regulatory audits routinely review supplier control records.
During MDSAP or FDA inspections, auditors frequently examine:
- Approved supplier lists
- Supplier evaluation records
- Risk-based justification for supplier classification
- Evidence of monitoring and corrective actions
Selecting a non-certified supplier increases documentation burden and may require deeper justification during audits.
Certification therefore acts as structural evidence of baseline compliance maturity.
How Certification Influences Medical Injection Molding Supplier Selection
For medical plastic injection molding suppliers, ISO 13485 impacts operational expectations at a detailed engineering level.
Process Validation Expectations

Manufacturers expect validated molding processes supported by:
- Installation qualification
- Operational qualification
- Performance qualification
- Defined process windows
- Capability studies such as Cp and Cpk
Without documented validation, variability in cavity pressure, temperature control, or cooling cycles can introduce regulatory risk.
For complex or small-scale components, especially in micro injection molding, process window stability becomes even more critical.
Tooling and Mold Control
Tool validation within mold making extends beyond dimensional checks. It includes:
- Cavity-to-cavity consistency verification
- Mold maintenance documentation
- Engineering change tracking
- Preventive maintenance records
Uncontrolled tooling changes are a frequent source of regulatory findings.
Where insert components are integrated into molded parts, as in medical insert molding, change control discipline becomes even more critical.
Material Traceability and Change Management
Medical grade resin must be traceable to certified raw material suppliers.
Effective control includes:
- Lot-level traceability
- Resin drying parameter documentation
- Regrind control policies
- Supplier change notification procedures
Certification demonstrates that such controls are embedded in the quality management system.
Cleanroom Integration and Documentation Flow
When cleanroom injection molding is combined with downstream medical device assembly and medical device packaging, environmental control becomes part of supplier evaluation.
ISO-certified suppliers typically integrate:
- Cleanroom monitoring procedures
- Personnel training records
- Environmental validation data
- Batch-level documentation suitable for device history records
SeaSkyMedical operates under an ISO-certified medical quality system with molding, assembly, and packaging performed in controlled cleanroom environments. This structure allows medical OEMs to incorporate supplier documentation directly into regulatory submissions without extensive remediation or secondary documentation reconstruction.
Certification and Inspection Intensity
ISO 13485 allows verification activities to be proportionate to supplier risk.
In practice:
- High-performing certified suppliers may qualify for reduced incoming inspection sampling.
- Lower-rated or non-certified suppliers often require expanded inspection plans.
This directly affects operational cost and production efficiency.
Certification therefore influences not only approval decisions but also downstream inspection workload.
Limitations of ISO 13485 Certification
Certification confirms the existence of a quality management system. It does not guarantee product conformity.
Manufacturers must still:
- Validate outsourced processes
- Conduct supplier audits when necessary
- Maintain risk management documentation
- Verify incoming product
Regulatory responsibility cannot be delegated.
However, certification reduces baseline uncertainty and narrows the scope of evaluation required during qualification.
Strategic Impact on Long-Term Supplier Partnerships
ISO 13485 certification affects supplier relationships beyond initial selection.
Certified suppliers are more likely to:
- Maintain structured corrective action systems
- Document engineering changes
- Provide audit-ready records
- Support regulatory inquiries
This predictability stabilizes long-term supply chains and reduces regulatory friction over time.
For medical device manufacturers seeking stable partners for injection molding and component production, certification functions as a measurable indicator of regulatory alignment and process discipline.
Conclusion
ISO 13485 certification influences supplier selection by shifting evaluation from price-based comparison to risk-based qualification.
It changes:
- Supplier risk classification
- Qualification cost
- Audit exposure
- Inspection intensity
- Documentation compatibility
While certification does not eliminate oversight, it reduces uncertainty and improves regulatory predictability within the supply chain.
Contact SeaSkyMedical for more information about ISO-certified medical injection molding and cleanroom manufacturing capabilities.




