The typical lead time for producing a new medical injection molded part is about 6 to 12 weeks after the design is released to the manufacturer.
This timeline usually covers mold design, tooling fabrication, mold trials, and the first dimensional inspection of molded parts produced through medical plastic injection molding.
Actual development time depends on part geometry, mold complexity, material selection, and validation requirements.
Understanding Lead Time in Medical Injection Molding
In manufacturing, lead time refers to the period between design release and the first approved production samples.

For projects involving medical plastic injection molding, this timeline includes both engineering work and mold fabrication. The process usually involves design evaluation, tooling development, mold trials, and inspection.
Medical projects may also include additional steps such as process validation, traceability documentation, and preparation for controlled manufacturing environments such as cleanroom injection molding.
Typical Timeline for Developing a Medical Injection Molded Part
Most medical injection molding projects follow a similar development schedule. Although each project is different, the typical development stages remain consistent.
| Development Stage | Typical Duration |
|---|---|
| Design review and manufacturability analysis | 1–2 weeks |
| Mold design | 1–2 weeks |
| Tooling fabrication | 3–5 weeks |
| Mold trials and process adjustment | 1–2 weeks |
| Inspection and validation | About 1 week |
Tooling fabrication typically represents the longest stage because the mold must be machined with extremely high dimensional precision.
Key Stages in the Medical Injection Molding Development Process
Design Review and Manufacturability Analysis
The development process usually begins with a design review and manufacturability analysis during early medical product development.
Engineers evaluate the CAD model to identify potential molding issues such as insufficient draft angles, uneven wall thickness, or complex undercuts.
Early design adjustments often reduce tooling complexity and prevent mold modifications later in the project.
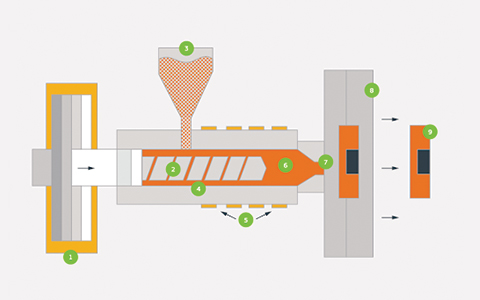
Medical Mold Design and Engineering
After the design review, engineers begin developing the injection mold structure using professional mold design services.
Typical mold design tasks include
- cavity and core layout
- runner and gate system design
- cooling channel configuration
- ejection system planning
For medical components with tight tolerances, the mold must maintain consistent dimensional stability throughout long production cycles.
Tooling Fabrication
Once the mold design is finalized, tooling fabrication begins through precision medical mold manufacturing.
This stage usually involves several machining processes including
- CNC rough machining of mold plates
- EDM machining for detailed cavities
- precision grinding and polishing
- mold assembly and fitting
Because medical molds often require hardened tool steel and precise surface finishes, tooling fabrication usually takes the largest portion of the overall development timeline.
Mold Trials and Process Optimization
After the mold is assembled, trial runs are conducted to verify the molding process.
Engineers evaluate the first molded parts and adjust process parameters such as injection pressure, melt temperature, cooling time, and mold temperature.
These trials help eliminate defects such as short shots, flash, sink marks, or part warpage. Multiple trial cycles may be required before the mold reaches stable production conditions.
Inspection and Validation
Once the molded parts meet dimensional requirements, the parts undergo inspection and process verification.

Inspection procedures commonly include
- dimensional measurements
- visual inspection
- functional evaluation
Medical projects may also require formal process validation to demonstrate consistent production performance across multiple molding cycles.
Factors That Influence Medical Injection Molding Lead Time
Although the typical lead time is about 6 to 12 weeks, several variables can affect the timeline.
Part Geometry and Design Complexity
Complex part geometries usually require more sophisticated molds.
Examples include
- thin wall medical components
- micro-scale features
- internal channels or undercuts
- tight dimensional tolerances
Small components produced through micro injection molding often require specialized tooling and extremely precise process control.
Mold Complexity and Cavity Count
The structure of the injection mold has a significant impact on development time.
Single-cavity molds are usually faster to manufacture, while multi-cavity molds require additional design and machining work.
Advanced manufacturing approaches such as medical insert molding or two shot injection molding may require more complex mold structures.
Medical Grade Material Selection
Material choice also affects development timelines.
Medical injection molding commonly uses materials such as
- polycarbonate
- polyethylene
- polypropylene
- PEEK
Disposable medical components are frequently produced using polypropylene injection molding due to its chemical resistance and cost efficiency.
Soft-touch medical components may require LSR injection molding or flexible elastomers for biocompatibility.
Validation and Manufacturing Requirements
Medical device components must meet strict manufacturing and quality standards.
Manufacturers may need to implement additional controls including
- cleanroom production preparation
- material traceability documentation
- process validation protocols
Many critical components are produced using cleanroom injection molding to ensure contamination control.
How Manufacturers Reduce Injection Molding Lead Time
Experienced manufacturers often use several engineering strategies to improve development efficiency.
Common approaches include
- performing early design for manufacturability analysis
- validating designs through 3D prototype printing
- optimizing mold cooling systems during design
- integrating automated inspection during mold trials
Early collaboration between product designers and manufacturers often helps prevent tooling redesign and reduces overall project timelines.
SeaSkyMedical works closely with customers during the early design phase to evaluate mold feasibility and identify potential manufacturing risks before tooling fabrication begins.
Situations That May Extend Development Time
Even well-planned injection molding projects may occasionally require additional engineering work.
Development timelines may increase when
- part designs are modified after tooling begins
- mold components require rework during trials
- materials are changed during validation
- additional testing is required for medical certification
Careful design evaluation early in the project helps reduce the likelihood of these delays.
Medical Components Commonly Produced Through Injection Molding
Injection molding is widely used to manufacture a broad range of medical components and disposable devices.
Typical examples include
- syringes and needle hubs
- IV connectors and tubing components
- diagnostic cartridges and test housings
- inhaler and drug delivery device components
- laboratory consumables such as pipette tips and specimen containers
Many of these products are manufactured as OEM medical components for global medical device companies.
Post Molding Assembly and Packaging
After molding, some medical products require additional manufacturing steps.

These secondary processes may include
- medical device assembly
- surface finishing
- ultrasonic welding
- sterile medical device packaging
These steps help prepare molded components for integration into finished medical devices.
Working With a Medical Injection Molding Manufacturer
Medical device companies often collaborate with specialized molding manufacturers to support both product development and production.
SeaSkyMedical is an ISO-certified medical injection molding company that provides end-to-end support for medical plastic components through medical device contract manufacturing.
The company assists with product design evaluation, mold development, precision molding, and controlled manufacturing processes.
Production uses certified raw materials and injection molding equipment in ISO cleanroom facilities followed by strict inspection procedures to ensure consistent product quality.
For more information about medical injection molding capabilities, you can contact SeaSkyMedical to discuss your project requirements.
FAQ
Q1 What is the typical lead time for producing a new medical injection molded part?
Most projects require about 6 to 12 weeks from design release to the first qualified molded parts. The timeline depends on mold complexity, material selection, and validation requirements.
Q2 Why does mold fabrication take the longest time in injection molding projects?
Injection molds require high precision machining and multiple finishing processes such as CNC machining, EDM machining, polishing, and assembly. These steps ensure dimensional accuracy and stable long-term production.
Q3 Can prototype tooling reduce development time?
Prototype molds made from aluminum or softer steel can shorten early development cycles and allow engineers to evaluate part performance before production tooling is built.
Q4 Does cleanroom production affect injection molding lead time?
Medical components that require cleanroom manufacturing may involve additional preparation and quality control procedures. These requirements can slightly extend the development timeline.
Q5 How can companies reduce lead time for injection molding projects?
Lead time can often be reduced through early design optimization, clear communication with manufacturers, and careful planning of materials, tooling design, and validation requirements.