Medical molding production line validation is a structured process used to confirm that equipment, materials, and manufacturing conditions can consistently produce compliant medical components.
It focuses on repeatability, traceability, and controlled variation across the entire production system, including early-stage design and product development services and downstream medical device contract manufacturing.
What Validation Means in a Medical Production Line Context
Validation is used to demonstrate that a production line can operate within defined limits and deliver stable output over time.
In a medical molding environment, this typically includes:
- Injection molding machines and tooling supported by mold making capabilities and medical plastic injection molding
- Cleanroom and environmental controls
- Material handling and storage systems
- Secondary operations such as assembly or packaging
- Inspection and measurement systems
Each element must be verified and linked through documentation.
Step-by-Step Validation Process for Medical Molding Production Lines
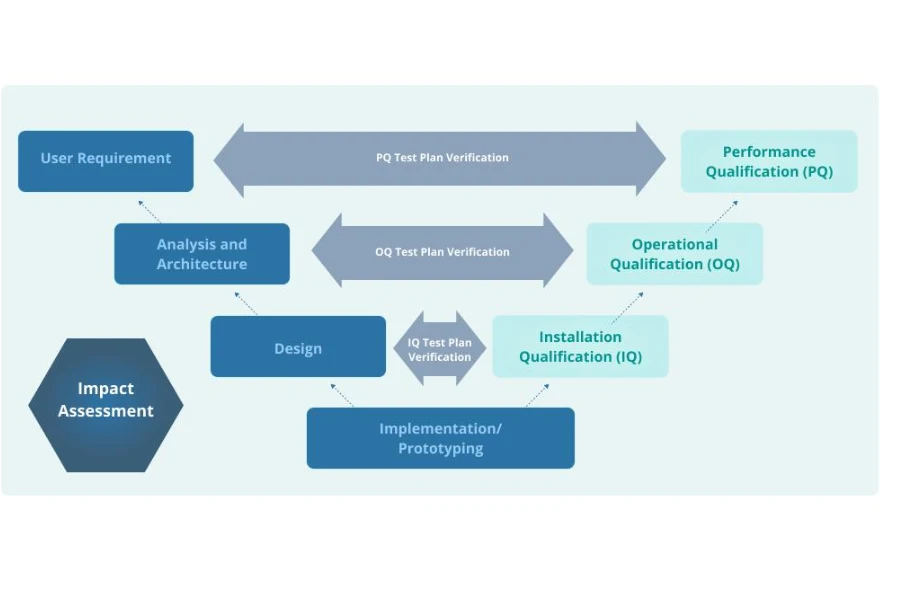
Validation is executed through a structured sequence. The goal is to move from installation verification to long-term production stability.
Step 1 Confirm installation and system readiness
This stage ensures that all equipment, utilities, and supporting systems are correctly installed and documented. Early validation planning often begins with 3D prototype printing and free mold tool design.
Step 2 Define and test process limits
The process is challenged across operating ranges to establish stable and controllable parameters.
Step 3 Demonstrate consistent production performance
The process is tested under routine production conditions to confirm repeatability across time, operators, and material batches.
This sequence aligns with the standard framework of IQ, OQ, and PQ.
Core Validation Framework Used in Medical Molding

Installation Qualification IQ
IQ verifies that the production line is installed according to design specifications and is ready for operation.
Typical IQ activities include
- Equipment verification including machine model, mold configuration, and auxiliary systems
- Utility checks such as electrical supply, compressed air, and cooling circuits
- Cleanroom verification including classification, airflow, and environmental controls
- Software and control system validation
- Documentation review including calibration, maintenance plans, and operating procedures
In molding environments, IQ often includes:
- Mold dimensional inspection and cavity verification supported by processes such as CNC plastic machining
- Thermal mapping of mold temperature systems
- Measurement system validation such as gage repeatability and reproducibility
IQ establishes the baseline condition for all subsequent testing.
Operational Qualification OQ
OQ determines whether the process can operate within defined limits and still produce acceptable parts.
It focuses on identifying a robust process window.
Key activities in OQ
- Functional testing of machine systems such as injection, clamping, and ejection
- Systematic variation of process parameters across high, low, and nominal conditions
- Dimensional and visual inspection at each parameter setting
- Identification of acceptable operating ranges
Critical parameters typically include:
- Melt temperature
- Injection speed
- Holding or pack pressure
- Cooling time
In practice, OQ often involves multiple runs across parameter combinations. This becomes more critical in multi-material processes such as 2K injection molding or medical insert molding.
A variation as small as a few degrees in melt temperature can shift material viscosity and affect fill consistency, especially in thin-wall medical parts.
OQ defines the operating boundaries that support stable production.
Performance Qualification PQ
PQ verifies that the process performs consistently under routine manufacturing conditions.
It introduces real-world variability.
Typical PQ activities include
- Running production batches using nominal parameters defined in OQ
- Including multiple operators and production shifts
- Using different raw material lots
- Sampling parts at defined intervals for inspection
Testing during PQ typically includes:
- Dimensional verification
- Functional performance checks
- Visual inspection
- Statistical evaluation of process capability
This is especially important for scaled manufacturing such as OEM medical components, where consistency across large volumes is required.
PQ confirms that the validated process can sustain performance over time.
Key Process Parameters That Drive Validation Outcomes

A validated molding process depends on controlling key variables that influence part quality.
Four parameters are commonly treated as primary drivers:
- Melt temperature
- Fill speed
- Pack pressure
- Cooling rate
These variables affect material flow, shrinkage behavior, and final part geometry.
Material behavior also depends on resin type, which is typically evaluated during material selection processes such as TPU injection molding.
Melt temperature is often the most difficult to measure directly. Inconsistent melt conditions can lead to variation between cavities or across production runs.
Stable monitoring and documentation of these parameters are required to maintain validation status.
Supporting Validation Systems in a Medical Molding Line
Validation extends beyond the molding process. Supporting systems must also be verified and controlled.
Cleanroom and Environmental Control Validation
Medical molding production often requires controlled environments, particularly in cleanroom injection molding environments.
Validation typically includes:
- Airborne particle monitoring
- Temperature and humidity control
- Airflow verification and pressure differentials
Continuous monitoring is required to maintain environmental consistency, especially in ISO-classified cleanrooms.
Material Validation and Traceability
Materials must be verified and traceable throughout the production process.
Validation activities include:
- Verification of medical-grade raw materials
- Lot traceability from incoming material to finished product
- Evaluation of compatibility with sterilization methods
Material variation between lots can affect processing behavior. OQ and PQ are designed to account for this variability.
Documentation and Data Integrity
Validation depends on complete and consistent documentation.
Typical records include:
- IQ, OQ, and PQ protocols and reports
- Device Master Record and Device History Record
- Material certifications and inspection data
Digital systems are increasingly used to maintain traceability across the full production lifecycle.
In practice, maintaining documentation alongside production activities reduces the risk of gaps during audits.
Inspection and Measurement System Validation
Measurement systems must be capable of detecting variation accurately.
Validation typically includes:
- Gage repeatability and reproducibility studies
- Calibration verification
- Alignment with product specifications and inspection methods
If measurement systems are not stable, process validation results may not be reliable.
Common Validation Challenges in Medical Molding
Even with a defined framework, validation can encounter practical issues.
Common challenges include:
- Difficulty measuring true melt temperature during processing
- Variation between material lots affecting viscosity and flow behavior
- Cavity imbalance in multi-cavity molds under edge conditions
- Measurement system drift over time
- Incomplete documentation linking process data to finished batches
Addressing these issues often requires coordination between engineering, quality, and production teams.
Advantages and Limitations of Production Line Validation
Advantages
- Provides evidence of consistent product quality
- Supports regulatory compliance and audit readiness
- Reduces process variation and unexpected defects
- Improves traceability across materials and production batches
Limitations
- Requires time and structured documentation effort
- Increases upfront validation workload
- Requires revalidation after significant process or equipment changes
- Depends on accurate measurement and process monitoring
Validation improves long-term stability but requires disciplined execution.
When Revalidation Is Required
A validated production line must be maintained over time.
Revalidation is typically required when:
- Equipment is relocated, replaced, or modified
- Mold design or tooling configuration changes
- Process parameters move outside validated ranges
- New materials or suppliers are introduced
- Environmental conditions or cleanroom classifications change
Risk-based assessment is used to determine the extent of revalidation required.
Integrating Validation into Medical Molding Operations
In practice, validation is not a separate activity. It is integrated into tooling development, process setup, and production control.
For example:
- Early design for manufacturability can reduce OQ iterations
- Coordinating mold design with process capability improves PQ outcomes
- Integrating downstream processes such as medical device assembly and medical device packaging simplifies validation across the full production line
- Managing post-molding steps through structured secondary operations improves consistency and traceability
Manufacturers that align tooling, molding, and controlled environment production within a unified system often reduce validation complexity and improve execution efficiency.
SeaSkyMedical applies this integrated approach across mold design, precision molding, and controlled manufacturing workflows, including advanced capabilities such as custom plastic molding and micro injection molding.
Conclusion
Medical molding production line validation is a structured process that demonstrates equipment readiness, process capability, and long-term production stability.
The combination of IQ, OQ, and PQ provides a framework for verifying that a production system can consistently produce compliant parts under controlled conditions.
When supported by environmental control, material traceability, and reliable measurement systems, validation becomes a practical tool for maintaining quality and reducing risk.
FAQ
Q1: How long does validation of a medical molding production line take
Most validation projects take between 3 and 6 months. The timeline depends on product complexity, number of cavities, and the extent of testing required during OQ and PQ.
Q2: What is the main difference between OQ and PQ
OQ defines the acceptable operating range of process parameters. PQ confirms that the process consistently produces acceptable parts under normal production conditions over time.
Q3: Is validation required for all medical molding production lines
Validation is required for most regulated medical products. The level of validation depends on device classification, application risk, and regulatory requirements.
Q4: Can validation results be reused for similar products
Partial reuse may be possible, but differences in geometry, material, or tooling often require additional validation or documented justification.
Q5: What is the most common issue during validation
Incomplete documentation and weak traceability are common issues. Even when parts meet specifications, missing records can lead to compliance failures.




