Traceability is a fundamental element of quality control in medical injection molding. Medical components must meet strict regulatory requirements and maintain consistent performance across production batches.
Manufacturers therefore track materials, process conditions, equipment usage, and inspection results throughout the manufacturing cycle. These traceability records connect each molded part to the production environment in which it was created.
This structured documentation allows manufacturers to verify product compliance, investigate quality deviations, and maintain repeatable production conditions.
What Traceability Means in Medical Injection Molding
Definition of Manufacturing Traceability
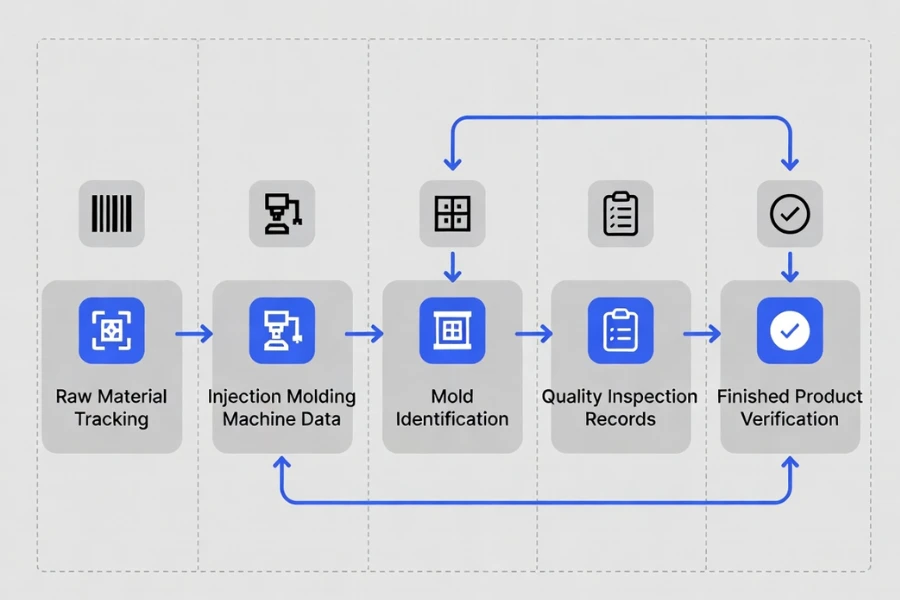
Traceability in medical injection molding refers to the ability to track materials, process parameters, equipment, and inspection data throughout the manufacturing lifecycle.
Each molded component can be linked to:
- the raw material batch used during production
- the molding machine and tooling involved
- the process parameters recorded during molding
- the inspection results verifying product quality
This connection between production data and finished parts forms the foundation of manufacturing quality control.
Why Traceability Is Required in Medical Device Manufacturing
Medical device manufacturing operates within strict regulatory environments. Manufacturers must demonstrate that products are produced under controlled conditions and meet defined quality standards.
Common regulatory frameworks include:
- ISO 13485 quality management systems
- FDA quality system regulations for medical devices
- European medical device regulations
Traceability allows manufacturers to show how a product was produced, inspected, and released for distribution.
Without documented traceability, manufacturers cannot reliably confirm compliance during audits or investigations.
What Production Data Must Be Traceable in Medical Injection Molding
Traceability systems track several categories of manufacturing data. These records allow engineers and quality teams to analyze production performance and confirm that molding processes remain within validated operating ranges.
Raw Material and Supplier Traceability

Material properties strongly influence molded part performance. Traceability systems therefore record the origin and certification of each polymer batch used in production.
Typical material records include:
- material grade and specification
- supplier identification
- material batch number
- certificate of analysis documentation
Material traceability is particularly important in medical applications because polymer composition can affect sterilization compatibility, chemical resistance, and long term mechanical stability.
Injection Molding Process Parameters

Injection molding quality depends heavily on controlled processing conditions. Manufacturers therefore monitor and record critical molding parameters during each production cycle.
Commonly tracked parameters include:
- melt temperature
- injection pressure
- holding pressure
- cooling time
- cycle time
Variations in these parameters can influence part shrinkage, internal stress, and dimensional accuracy. Traceability records allow engineers to confirm that production remained within validated process windows.
Molding Machine and Tooling Identification
Injection molding machines and molds also affect part quality. Traceability systems therefore record which equipment was used during production.
Typical equipment identification includes:
- molding machine identification
- mold number and cavity location
- tooling maintenance history
- mold cycle counts
These records help manufacturers detect quality deviations related to equipment wear or maintenance conditions.
Production Batch Records
Medical injection molding commonly uses batch based production tracking. Each production run receives a lot number that links parts to their manufacturing history.
Batch records may include:
- lot number identification
- production date and shift
- operator identification
- material batch used during production
These records provide a clear reference point when quality issues must be investigated.
Inspection and Quality Verification Records
Quality inspection is another key component of traceability systems. Inspection results confirm that molded components meet dimensional and functional requirements.
Inspection documentation typically includes:
- dimensional measurement results
- visual inspection records
- functional verification tests
- process monitoring data
These records demonstrate that production batches meet defined quality specifications.
How Traceability Improves Quality Control in Medical Injection Molding
Traceability directly supports quality control by connecting product performance with manufacturing conditions. This connection allows engineers to identify process deviations and maintain consistent production quality.
Faster Root Cause Identification
When a defect is detected, traceability records allow engineers to review the production history of affected parts.
Investigations often examine:
- material batch information
- molding process parameters
- machine and tooling identification
This structured data helps engineers locate the source of variation more quickly than manual investigation methods.
Prevention of Recurring Manufacturing Defects
Historical traceability records reveal patterns in production performance. Engineers analyze these records to identify unstable process parameters or recurring equipment conditions.
Corrective actions may include:
- adjusting process parameters
- modifying tooling maintenance schedules
- refining process validation limits
This analysis helps reduce the likelihood of recurring defects.
Improved Process Consistency
Injection molding processes rely on repeatable production conditions. Traceability systems allow manufacturers to confirm that process parameters remain consistent across production runs.
Maintaining stable molding conditions helps ensure:
- dimensional repeatability
- uniform mechanical properties
- consistent surface finish quality
These factors are essential in medical device manufacturing where tolerances are tightly controlled.
Controlled Product Recall Management
Traceability systems also support product recall management. If a quality issue affects specific batches, manufacturers can identify which products were produced under the same conditions.
This approach allows manufacturers to isolate affected batches rather than recalling all products in circulation. Targeted recalls reduce supply disruption while maintaining patient safety.
Traceability Systems Used in Medical Injection Molding
Manufacturers use several methods to manage production traceability depending on regulatory requirements and product risk levels.
Lot Based Traceability Systems
Lot tracking is widely used in manufacturing environments. Parts produced during the same production run share a common lot number.
Lot tracking allows manufacturers to connect finished products with:
- material batches
- production dates
- inspection records
This approach is efficient for managing traceability across high volume production runs.
Serial Number Traceability
Some medical products require unit level traceability. Each component receives a unique serial number that records its individual manufacturing history.
This method allows detailed tracking of individual devices but requires more extensive data management systems.
Digital Traceability and Manufacturing Data Systems
Modern medical manufacturing facilities increasingly rely on digital production systems to manage traceability data.
Examples include:
- manufacturing execution systems
- automated machine data collection
- electronic production records
Digital traceability systems allow engineers to analyze production data and identify process trends more efficiently.
Challenges in Implementing Manufacturing Traceability
Although traceability improves quality control, implementing these systems introduces operational challenges.
Manufacturing Data Integration
Manufacturing data may originate from multiple sources such as molding machines, inspection equipment, and quality management systems. Integrating these data sources into a unified traceability framework requires careful system planning.
Managing Large Volumes of Production Data
Injection molding processes generate large amounts of process and inspection data. Manufacturers must maintain structured data storage systems that allow records to be retrieved efficiently during audits or investigations.
Balancing Traceability and Operational Complexity
Traceability systems increase documentation and data management requirements. Manufacturers therefore apply risk based strategies to determine the appropriate level of traceability for each product type.
How Medical Injection Molding Manufacturers Implement Traceability
Traceability is typically integrated into the manufacturing workflow of medical injection molding facilities.
A common traceability workflow includes:
1 verification of incoming raw materials
2 assignment of material batch identification
3 identification of molding machines and molds used in production
4 recording of injection molding process parameters
5 in process inspection and measurement
6 final quality documentation and batch release
Manufacturers such as SeaSkyMedical integrate controlled documentation practices and cleanroom production environments to maintain consistent traceability throughout medical molding operations.
These systems connect material records, process monitoring data, and inspection results within the overall quality management system.
Conclusion
Traceability plays a central role in maintaining quality control in medical injection molding. By linking materials, manufacturing conditions, and inspection records, traceability systems provide visibility into the production process.
These records support regulatory compliance, enable faster defect investigation, and help manufacturers maintain stable production conditions. As medical manufacturing becomes increasingly data driven, traceability systems will remain essential for ensuring product reliability and patient safety.
For more information about controlled medical injection molding processes, contact SeaSkyMedical.
FAQ
Q1 What is traceability in medical injection molding
Traceability refers to the ability to track materials, equipment, process parameters, and inspection data throughout the manufacturing lifecycle. These records allow manufacturers to verify product quality and identify the origin of production issues.
Q2 Why is traceability important in medical device manufacturing
Medical devices must meet strict regulatory requirements. Traceability ensures that manufacturers can document how products were produced and demonstrate compliance during quality audits or product investigations.
Q3 What information is typically tracked in medical injection molding
Manufacturers commonly track material batches, molding parameters, machine identification, production lot numbers, and inspection results. These records provide a complete manufacturing history for each production batch.
Q4 How does traceability support defect investigation
Traceability records allow engineers to review the production history of affected parts. By analyzing material data, equipment records, and process parameters, manufacturers can identify the root cause of quality deviations.
Q5 Does traceability increase manufacturing complexity
Traceability systems require additional documentation and data management. However, these systems improve quality control, regulatory compliance, and risk management in medical manufacturing environments.




