Introduction: Why Dual-Chamber Syringes Are the Preferred Choice for Complex Medications
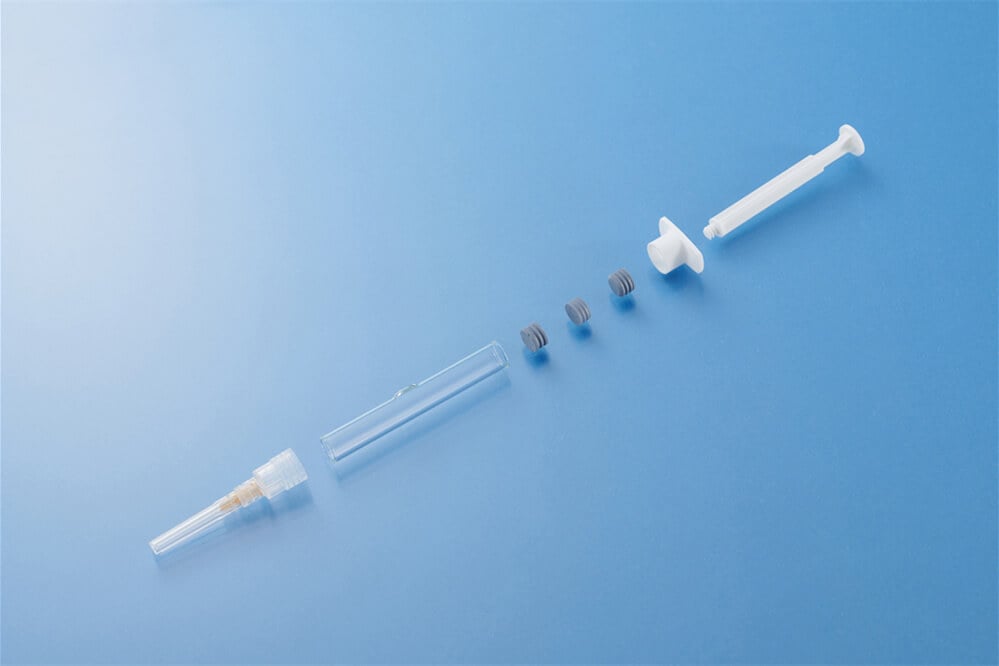
Administering biologics, vaccines, hormones, and coagulation factors poses unique challenges because these therapies are often unstable in liquid form and highly sensitive to environmental conditions such as temperature and humidity. Traditional vial-and-diluent kits involve multiple preparation steps, increasing contamination risk and making precise dosing difficult, which can create obstacles both for healthcare professionals and patients. Dual-chamber syringes, also referred to as dual-chamber devices (DCDs) or dual-compartment injection systems, integrate separate chambers for a lyophilized drug and a diluent or for two incompatible liquids, maintaining drug stability during storage and enabling on-demand mixing or sequential delivery. As regulatory agencies including the FDA and EMA recognize their benefits and as market adoption continues to accelerate, these devices have become essential in modern injectable drug delivery, with production standards increasingly aligned with precision medical device contract manufacturing capabilities provided by companies like SeaSkyMedical.
Basic Structure and Mechanism of Dual-Chamber Syringes
Concentric Design
In the classical concentric configuration, two chambers are aligned along a single axis, with the front chamber near the needle tip typically containing the lyophilized drug or one liquid and the rear chamber holding the diluent or second liquid. A movable middle plunger separates the two compartments, while a bypass channel enables fluid transfer when pressure is applied. During administration, pressing the plunger compresses the rear chamber, driving the liquid through the bypass to mix with the front chamber’s contents, which then proceeds to the needle for delivery. This design accommodates both liquid/powder reconstitution and liquid/liquid formulations that require sequential or simultaneous delivery. While widely implemented and highly mature, concentric designs may exhibit variable activation force across different plunger stages, extended stroke length requiring two-handed operation, and elevated manufacturing complexity due to precision bypass structures that rely on advanced mold making and free mold tool design technologies.
Lateral-Chamber Design
Emerging lateral-chamber configurations position the two compartments side by side rather than coaxially, connected through a narrow channel with a one-way valve to prevent backflow. Reconstitution involves a simple rotational or suction action transferring diluent into the drug chamber, followed by standard plunger actuation to deliver the medication. This configuration maintains consistent glide force throughout the stroke, enhancing dose reliability, supporting single-handed operation, simplifying manufacturing, and reducing production costs, while minimizing packaged height for improved storage and transport. Variants include ampoule-integrated systems with built-in breakers and microfilters and ampoule-free designs suitable for larger volumes or integration with autoinjectors.
Design Comparison and Selection Guidance
| Feature | Concentric Design | Lateral-Chamber Design |
|---|---|---|
| Maturity | Established, widely marketed | Emerging, some in development |
| Ease of Use | Moderate (requires adaptation to bypass stroke) | High (similar to standard prefilled syringes) |
| Manufacturing Cost | Higher | Lower |
| Dose Accuracy | Sensitive to activation variability | Stable and consistent |
| Volume Capacity | Typically ≤3 mL | Up to 20 mL |
| Suitable Applications | Standard reconstitution needs | Large-volume delivery, autoinjectors, high usability requirements |
Core Advantages of Dual-Chamber Syringes
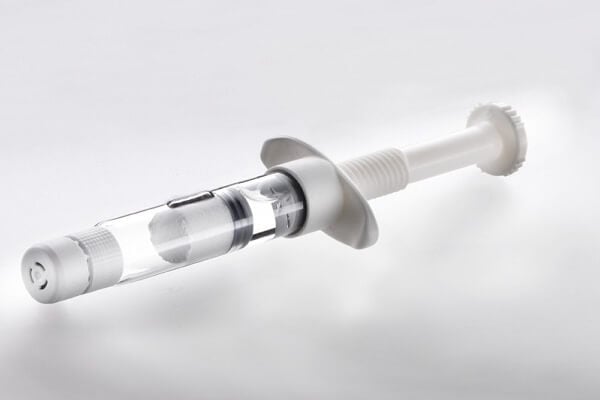
Enhanced Drug Stability
By maintaining separate chambers for lyophilized drugs and diluents or incompatible liquids, dual-chamber syringes prevent hydrolytic and chemical degradation, extend shelf life, and reduce reliance on cold chain storage, particularly for temperature-sensitive biologics and complex formulations.
Simplified Operation Supporting Home Use
Compared with traditional multi-component kits, dual-chamber devices reduce the number of preparation steps by approximately fifty percent, enabling patients or caregivers to mix and inject medication in a single, streamlined action. Surveys indicate high patient satisfaction in chronic disease populations such as hemophilia and diabetes, reflecting the growing trend toward hospital-at-home and self-administration models.
Cost Reduction and Waste Minimization
By eliminating overfill requirements, minimizing post-opening drug waste, and simplifying inventory management, dual-chamber syringes reduce both direct and indirect healthcare costs. Single-dose designs further enhance safety and efficiency across supply chains, while companies like SeaSkyMedical ensure precision manufacturing reduces variability and overall production waste.
Rapid Response in Emergencies
For acute care scenarios, including administration of glucagon for severe hypoglycemia or chemical antidotes, dual-chamber syringes enable immediate drug reconstitution and delivery, significantly shortening preparation time and enhancing clinical responsiveness.

Key Applications and Marketed Products
Hormones and Hematologic Disorders
- Growth hormone: Genotropin (Pfizer), Humatrope (Eli Lilly), Saizen (Merck Serono)
- Coagulation factor VIII: Xyntha Solofuse (Pfizer)
- Erythropoietin: Mircera (Roche)
- Parathyroid hormone: Preotact (Nycomed)
Diabetes
- Exenatide suspension: Bydureon, Byetta (AstraZeneca)
- Albiglutide freeze-dried formulation: Tanzeum (GSK)
- Glucagon rescue pens for severe hypoglycemia (under development)
Oncology and Autoimmune Disease
- Leuprolide: Lupron Depot (Abbott)
- Aripiprazole: Abilify Maintena (Otsuka)
- Pegylated interferon α-2b: PegIntron (Sagent/Selexis)
Vaccines and Infectious Diseases
- Conjugate vaccines: PRP-T + DTP-IPV
- ViVAXIM (typhoid-hepatitis A combination)
- Measles-rubella: reduced open vial wastage
- Dual-chamber powder-liquid vaccines for emergency deployment
Manufacturing and Quality Control Challenges
Lyophilization Specifics
Dual-chamber devices’ elongated geometry and limited contact areas make freeze-drying more complex than conventional vials. Aluminum block holders provide uniform heat transfer, whereas organic glass holders rely on radiation, impacting drying homogeneity. Continuous freeze-drying technologies, such as the “DCC LyoMate” system, allow real-time monitoring and improve batch consistency. Advanced production often involves CNC plastic machining and secondary operations including precise medical device packaging to maintain sterility and function.
Container Closure Integrity (CCI)
Movable components including middle plungers and needle shields complicate CCI testing. Methods such as vacuum decay, dye ingress, and high-voltage leak detection are necessary to ensure long-term seal integrity.
Extractables & Leachables (E&L)
Potential sources include:
- Glass: tungsten, aluminum, barium ions
- Rubber plunger: antioxidants, vulcanization agents
- Silicone oil: risk of protein aggregation
- Adhesives: residual solvents
Mitigation strategies involve low-extractable polymers (COP/COC), fluoropolymer-coated plungers, tungsten-free processes, silicone-free designs, and comprehensive E&L analysis. SeaSkyMedical leverages material selection expertise to optimize component safety and device performance.
Material Selection: Glass vs Plastic
Glass
While glass provides excellent transparency, gas barrier properties, and cost-effectiveness, it carries risks of breakage, tungsten residues, metal ion leaching, and micro-particles at the glass-rubber interface.
Plastic (COP/COC/CZ)
Plastics offer safety from breakage, precise dimensions, lightweight ergonomics, low extractables, and silicone-free options. Manufacturing relies on TPE injection molding, medical plastic injection molding, and cleanroom injection molding techniques to maintain dimensional accuracy and sterility.
Silicone Considerations
Silicone oil ensures smooth plunger movement but can induce aggregation in sensitive proteins. Alternatives include fluoropolymer coatings, chemical vapor deposition, plasma treatments, and silicone-free pistons, reducing protein interaction risk while maintaining operational reliability.
Frequently Asked Questions (FAQ)
What distinguishes a dual-chamber syringe from a conventional prefilled syringe?
Dual-chamber syringes contain two independent compartments, mixing contents only at the time of use, significantly extending the shelf life of unstable biologics compared with single-chamber prefilled syringes.
Which drug types are suitable for dual-chamber syringes?
They are suitable for biologics, hormones, coagulation factors, vaccines, and formulations that cannot be co-formulated due to incompatibility.
Is patient operation complicated?
Devices are designed for simplicity, typically requiring a single press or a simple draw. Minimal instruction is sufficient for elderly patients or self-injectors.
Are there risks of glass particulates or tungsten contamination?
Current devices mitigate these risks through plastic materials, tungsten-free processes, inline filters, and strict E&L control measures.
Does silicone oil affect protein drugs?
Silicone can induce aggregation in sensitive proteins; however, silicone-free designs with specialized coatings or pistons offer a safe alternative.
Is the dosage accurate?
Accurate dosing is achievable if proper technique is followed, including complete transfer of diluent and removal of air bubbles. Lateral-chamber designs further enhance reliability.
Can dual-chamber syringes be used with autoinjectors?
Yes, several autoinjector products incorporate dual-chamber designs. Lateral-chamber configurations provide mechanical reliability for sequential or high-volume delivery.
Conclusion and Future Outlook
Dual-chamber syringes have become the preferred delivery system for complex injectable medications by combining stability, simplified operation, and reduced waste. Innovations in plastic materials, lateral-chamber configurations, silicone-free designs, and continuous freeze-drying technology address previous limitations, while the expanding demand for home healthcare and novel biologics fuels market growth. With an anticipated CAGR of 6–8%, early integration of dual-chamber systems in product development enables manufacturers to optimize lifecycle management, improve patient adherence, and enhance brand differentiation, ensuring critical medications are delivered safely, efficiently, and reliably.




