Material traceability in medical injection molding is not optional. It is driven by regulatory expectations that require manufacturers to identify and document materials used in finished medical devices produced in regulated medical plastic injection molding environments.

Under 21 CFR Part 820, manufacturers must establish procedures for identification and traceability where appropriate. Sections 820.60 and 820.65 define requirements for product identification and traceability, particularly for devices where failure could result in serious harm.
ISO 13485 further requires documented control of purchased product and maintenance of Device History Records. These records must link raw material lots to production batches and final release status.
For implantable or life-sustaining devices, traceability granularity is typically tighter. Material lot identification must be maintained through molding, assembly, and packaging without interruption.
Traceability therefore begins at raw resin receipt and extends through final device shipment.
How Material Traceability Changes Production Architecture
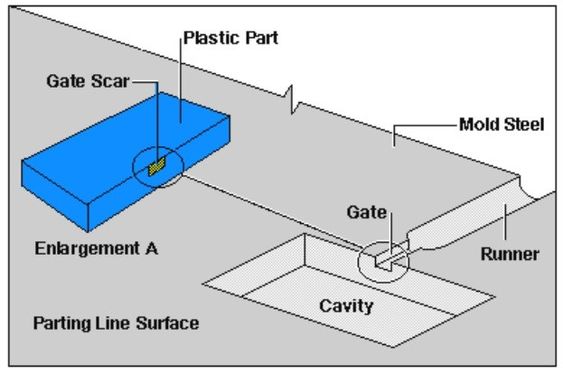
Strict material traceability reshapes how a medical injection molding facility structures its production system.
It affects lot sizing, scheduling logic, changeover frequency, and documentation workload.
In a regulated environment, production is often organized around material lots rather than purely around volume efficiency.
If a resin lot is exhausted mid-run, a documented batch break is required before introducing a new lot. This increases:
- Line clearance activities
- Label verification steps
- Documentation entries
- Potential downtime
Lot-based production planning may reduce average run length. While this can lower overall equipment efficiency, it improves containment precision.
Traceability therefore shifts production strategy from maximum throughput to controlled segmentation.
Raw Material Control and Segregation Strategy
Material traceability begins during material selection and continues at receiving.
Each resin lot is:
- Logged with supplier identification
- Verified against Certificate of Analysis
- Assigned internal trace code
- Released or quarantined
In medical molding, mixing resin lots to optimize inventory turnover is typically avoided. Segregation reduces variability and simplifies root cause analysis.
For hygroscopic resins such as polycarbonate or nylon, drying parameters must be recorded per material lot. Moisture variation between lots can affect melt viscosity and dimensional stability.
Drying logs are commonly tied to lot identification to maintain process consistency.
Regrind usage is often restricted or eliminated in regulated medical parts. Where permitted, regrind must remain traceable to its original material lot and production batch.
Impact on Validation and Process Capability
Material traceability directly influences process validation strategy.
During IQ, OQ, and PQ activities, validation is conducted using defined material lots. Process windows are established based on that material’s behavior.
This becomes particularly critical in high-precision applications such as micro injection molding, where dimensional tolerance sensitivity is higher.
If a new supplier or resin formulation is introduced, manufacturers must evaluate whether revalidation is required.
Traceability enables correlation between:
- Material lot
- Machine parameter setpoints
- In-process inspection results
- Cpk and dimensional trends
If process capability shifts over time, material lot comparison becomes part of the investigation.
Without material traceability, statistical variation cannot be confidently attributed to equipment, tooling, or resin variability.
Traceability therefore supports both compliance and engineering analysis.
Cleanroom Operations and Material Handling Controls
In ISO 7 or ISO 8 cleanroom injection molding environments, material handling discipline is linked to traceability.
Common controls include:
- Controlled staging areas for released material
- Lot-labeled containers within cleanroom
- Documented material reconciliation per shift
- Clear separation of approved and nonconforming stock
Material dwell time in controlled environments may also be monitored. Extended exposure for certain polymers can influence moisture uptake and processing stability.
Traceability in cleanroom settings therefore combines contamination control with lot accountability.

Operational Constraints Introduced by Strict Lot Control
Strict traceability introduces operational complexity.
Production constraints may include:
- Smaller production segments
- Increased batch breaks
- Higher documentation workload
- Reduced flexibility in material substitution
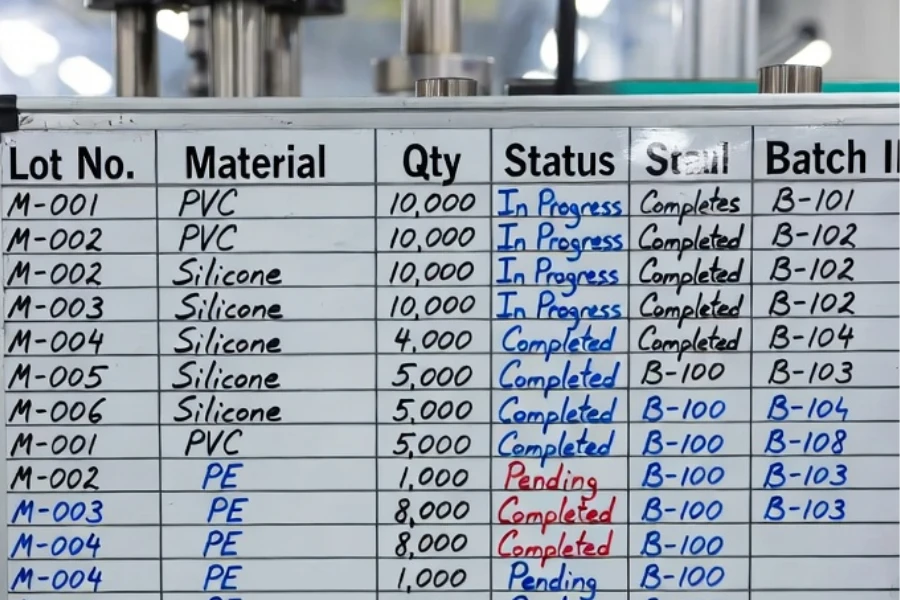
Inventory fragmentation can increase working capital requirements. ERP or MES systems are often required to maintain real-time linkage between material, machine, and batch data.
These constraints represent trade-offs rather than disadvantages. Manufacturers balance operational efficiency against regulatory risk containment.
Risk Containment and Recall Precision
Material traceability determines recall scope.
If a material-related issue is identified, traceability allows isolation of only the affected production batches.
Without lot linkage, recall scope may expand to all devices produced within a broader timeframe.
The financial and reputational impact difference can be substantial.
Traceability therefore functions as a risk containment mechanism rather than simply a compliance requirement.
Practical Trade-Offs in Real Manufacturing Environments
Material traceability increases administrative workload and production segmentation.
However, it reduces uncertainty during audits, investigations, and field performance reviews.
Manufacturers must weigh:
| Factor | With Strict Traceability | Without Structured Traceability |
|---|---|---|
| Recall scope | Narrow and controlled | Broad and costly |
| Root cause speed | Faster identification | Extended investigation |
| Documentation load | Higher | Lower |
| Operational flexibility | Reduced | Higher |
In regulated medical environments, structured traceability is typically considered necessary despite operational impact.
How SeaSkyMedical Implements Structured Material Traceability
SeaSkyMedical operates under ISO 13485 quality management principles and maintains documented linkage between raw material lots and production batches across its medical device contract manufacturing operations.
Material receipt verification, controlled lot release, and cleanroom-segmented production records are integrated into standard operating procedures.
Production, medical device assembly, and medical device packaging are conducted in ISO-classified cleanroom environments where material segregation and documentation controls are enforced.
In multi-material processes such as medical insert molding, traceability extends beyond resin to include embedded components and secondary materials.
Traceability at SeaSkyMedical is implemented as a production architecture decision rather than a reactive compliance measure.
For manufacturers evaluating structured medical injection molding systems, Contact SeaSkyMedical for more information or to request a quotation.