Material traceability affects medical production by directly controlling regulatory compliance, process consistency, and risk containment across the entire manufacturing lifecycle, since every deviation in material origin or condition can propagate through processing and ultimately influence device performance and patient safety.
What is material traceability in medical production

Material traceability in medical production refers to the ability to systematically record and connect the origin, identity, processing conditions, and movement of materials and components throughout the entire lifecycle of a medical product, allowing manufacturers to reconstruct both upstream material sources and downstream product distribution with precision.
In medical environments, this extends beyond finished product identification and includes raw material batch numbers, supplier certifications, processing parameters such as drying conditions, melt temperatures, and pressures, as well as inspection results, equipment identifiers, and operator records, forming a continuous data structure that links material behavior to final product performance.
It is also necessary to distinguish between tracking and traceability, since tracking focuses on where a product is located at a given time, while traceability explains how that product was produced and which materials and conditions were involved, which is the basis for both regulatory validation and engineering analysis.
Why material traceability is critical in medical manufacturing
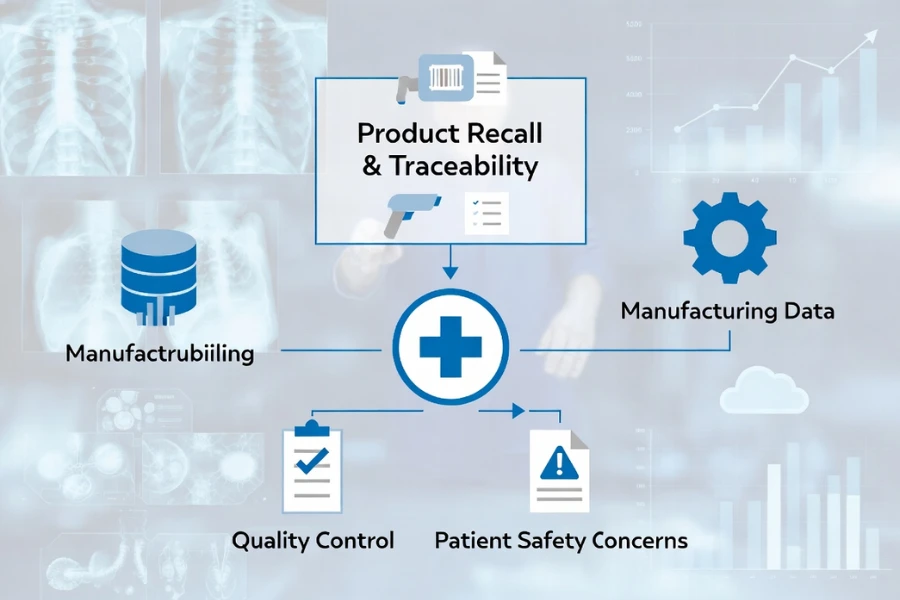
Material traceability is a foundational control mechanism in medical manufacturing because it determines whether a product can be proven compliant, reproducible, and safe under real production conditions rather than theoretical specifications.
From a regulatory perspective, traceability is required by frameworks such as ISO 13485, FDA quality system regulations, and EU MDR, all of which require documented relationships between materials, processes, and finished devices, meaning that without traceability, compliance cannot be demonstrated or audited.
From a safety standpoint, traceability allows manufacturers to isolate defective material batches with precision, enabling targeted recalls instead of full product withdrawals, which significantly reduces patient exposure and operational disruption.
From a process control perspective, traceability enables manufacturers to link production outcomes to specific material conditions, allowing consistent replication of validated processes, which is essential in high-tolerance medical components where small variations can lead to functional failure.
This is not only a documentation requirement — it is a process control requirement, because without material traceability, manufacturing variability cannot be explained, controlled, or improved.
Regulatory requirements for material traceability
Material traceability in medical manufacturing is defined through structured regulatory frameworks that require manufacturers to maintain auditable connections between materials, processes, and product performance across the full lifecycle.
ISO 13485 traceability requirements
ISO 13485 requires organizations to establish and maintain traceability procedures that cover materials, components, and environmental conditions when they can influence safety or performance, with additional requirements for implantable devices where full material identification must be retained.
FDA traceability and material control
Under FDA regulations, including 21 CFR 820, traceability is embedded within the quality system through requirements for design control, process validation, and material identification, while materials must remain traceable to approved sources and documented processing conditions.
The implementation of Unique Device Identification strengthens traceability by linking each device to production data such as batch numbers and manufacturing records, allowing regulators and manufacturers to connect finished products back to material inputs.
EU MDR lifecycle traceability
The EU MDR framework requires lifecycle traceability across design, production, and post-market stages, while extending traceability responsibilities across the entire supply chain, ensuring that material and process data remain connected beyond manufacturing.
Material verification and biocompatibility
Standards such as ISO 10993 require traceable documentation of biocompatibility testing, ensuring that material performance and safety can be verified against specific batches used in production.
How material traceability affects medical production processes
Material traceability directly shapes how medical production processes are controlled, validated, and optimized, because material variability is one of the primary sources of manufacturing deviation.
Traceability in injection molding operations
In medical plastic injection molding, each resin batch is associated with supplier documentation and must be processed under controlled conditions, where parameters such as drying temperature, time, and residual moisture content are recorded to prevent defects such as bubbles, voids, or surface streaking.
If material moisture is not properly controlled, even small deviations can lead to internal defects that are not immediately visible but may compromise mechanical strength or sealing performance, particularly in thin-walled components often produced through micro injection molding.
During molding, parameters such as melt temperature, injection pressure, and cooling rates are linked to specific material batches, allowing manufacturers to identify how variations in material properties influence shrinkage, warpage, or dimensional stability, which are often addressed through precise mold making strategies.
Material variation leads to process variation, and process variation leads to product variation, which is why traceability must connect these stages rather than treat them as separate data points.
Cleanroom environment traceability
In controlled cleanroom injection molding environments, traceability extends beyond materials to include environmental conditions such as particle levels, temperature, and humidity, since contamination risks and material behavior are influenced by these variables.
Automated data capture systems are typically used to integrate environmental data with material and process records, reducing manual intervention while maintaining consistent production quality.
Post-processing and sterilization linkage
Traceability continues through post-processing, including medical device assembly and medical device packaging, where parameters such as sterilization method, exposure time, temperature, and dosage are recorded and linked to material batches and product lots.
This linkage is critical because material performance may change after sterilization, and without traceability, it is not possible to determine whether deviations originate from materials or downstream processes.
Internal and external supply chain traceability
Internally, traceability connects material receipt, inspection, storage, production, and packaging, while externally it links suppliers, logistics providers, and distributors, forming a continuous data chain that reflects both physical material flow and associated data flow.
Serialization and aggregation
Serialization assigns unique identifiers to individual products, while aggregation links these identifiers to packaging hierarchies, enabling efficient tracking and verification throughout distribution channels.
In practice, this integrated traceability approach is typically implemented within structured medical device contract manufacturing environments where material control, process monitoring, and documentation are aligned.
Risks of missing material traceability
The absence of material traceability introduces systemic risks because it removes the ability to isolate, analyze, and control manufacturing variables.
Without traceability, defects cannot be linked to specific material batches, which often results in broad recalls that increase cost and extend patient exposure to risk.
Regulatory consequences include audit failures, warning notices, and loss of market access, while financial impacts can include recall costs, production downtime, and legal liabilities.
More importantly, without traceability, root cause analysis becomes speculative rather than evidence-based, which increases the likelihood of repeated failures and undermines long-term process stability.
How traceability systems are implemented
Material traceability systems are implemented through a combination of identification methods, data integration platforms, and automated data collection processes that ensure consistent and reliable recording of production information.
Identification and marking technologies
Technologies such as laser marking, Data Matrix codes, barcodes, and RFID tags are used to assign unique identifiers to materials and products, enabling traceability across production and distribution.
Data collection and system integration
Manufacturing Execution Systems capture real-time production data, while Product Lifecycle Management and Application Lifecycle Management systems integrate product, process, and compliance information into a unified traceability structure.
Automation and IoT-enabled monitoring
Automation reduces manual input errors by enabling automatic data capture, while IoT sensors monitor environmental and transportation conditions, ensuring that material integrity is maintained across the supply chain.
Integrated traceability platforms
Advanced platforms connect internal production data with external supply chain information, creating a digital representation of material flow and process history that supports real-time visibility and decision-making.
Challenges and practical considerations
Implementing material traceability introduces operational challenges related to cost, data complexity, and supplier variability, particularly in high-precision medical manufacturing environments.
High-performance materials require strict control and detailed documentation, which increases system complexity, while small or complex components may require specialized marking methods that do not affect functionality.
Supplier inconsistency can create gaps in traceability data, requiring additional validation and documentation, while large volumes of data must be securely stored and easily retrievable to meet regulatory expectations.
To address these challenges, manufacturers typically adopt structured approaches such as early-stage product development alignment, rapid validation through 3D prototype printing, and integrated process control within custom plastic molding environments.

Future trends in traceability systems
Material traceability is evolving toward more connected and predictive systems that provide real-time visibility and advanced data analysis capabilities.
IoT technologies enable continuous monitoring of material and environmental conditions, while digital integration across supply chains improves transparency and responsiveness.
Emerging approaches such as blockchain are being explored to enhance data integrity, while artificial intelligence is increasingly used to analyze traceability data and identify patterns that may indicate potential risks before they result in product failures.
Digital supply chain models further extend this capability by enabling simulation and predictive planning, allowing manufacturers to anticipate disruptions and optimize production strategies.
Conclusion
Material traceability is a core control mechanism in medical production that links materials, processes, and products into a verifiable system capable of supporting compliance, quality consistency, and risk management.
By establishing clear relationships between material inputs and production outcomes, manufacturers can reduce variability, improve process control, and respond effectively to quality issues, which is essential in regulated medical environments.
In practice, implementing traceability requires integrated control of materials, processes, and environments within structured manufacturing systems such as those used in OEM medical components production.
Contact SeaSkyMedical for more information on implementing material traceability in medical manufacturing.
FAQ
Q1: What does material traceability mean in medical production?
Material traceability refers to the ability to track materials from their origin through manufacturing and distribution, linking each batch to processing conditions, inspection results, and final product use, so that any issue can be traced back to its source and verified with documented evidence.
Q2: Why is material traceability required in medical manufacturing?
Material traceability is required to meet regulatory standards such as ISO 13485, FDA regulations, and EU MDR, while also enabling precise recall management, risk control, and verification of product safety and performance, which are essential for market approval and patient protection.
Q3: How does UDI relate to material traceability?
UDI provides a unique identifier for each medical device, linking finished products to production data such as batch numbers and manufacturing records, which allows traceability systems to connect device-level information back to material batches and processing conditions.
Q4: How can small medical components achieve traceability?
Small components can be traced using methods such as laser marking or micro Data Matrix codes, or by linking items to packaging-level identifiers through aggregation, ensuring that each unit can be associated with its material and production data without affecting functionality.
Q5: How can manufacturers handle missing supplier traceability data?
Manufacturers typically address this by establishing supplier quality agreements, requiring batch documentation and certifications, performing incoming material verification, and prioritizing suppliers with established quality systems to ensure consistent and reliable traceability data.
Q6: Does material traceability significantly increase production cost?
Traceability systems require initial investment in equipment, software, and training, but the long-term benefits such as reduced recall scope, improved compliance, and enhanced process control generally outweigh the cost, making traceability a necessary and practical investment in medical manufacturing.